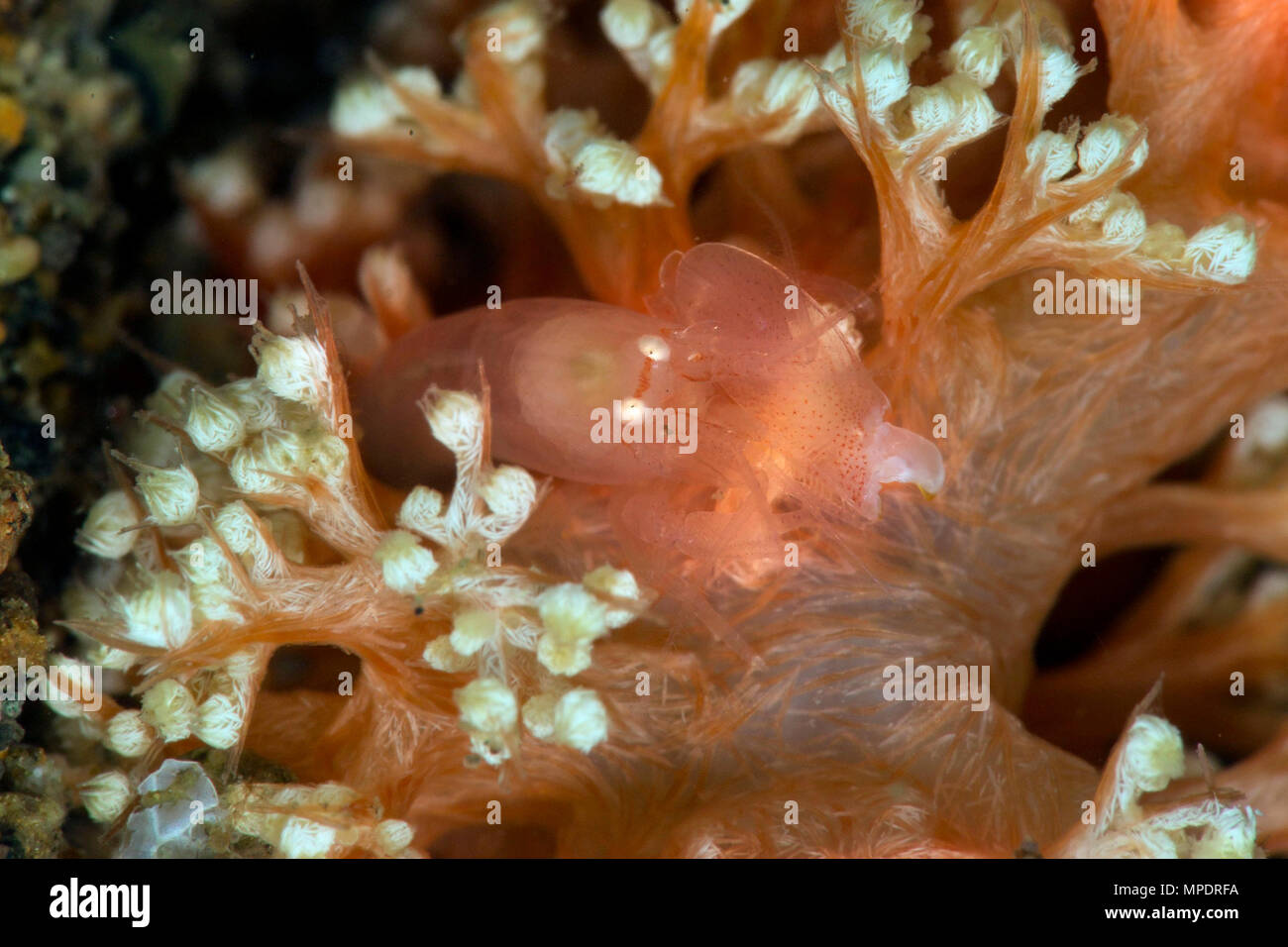
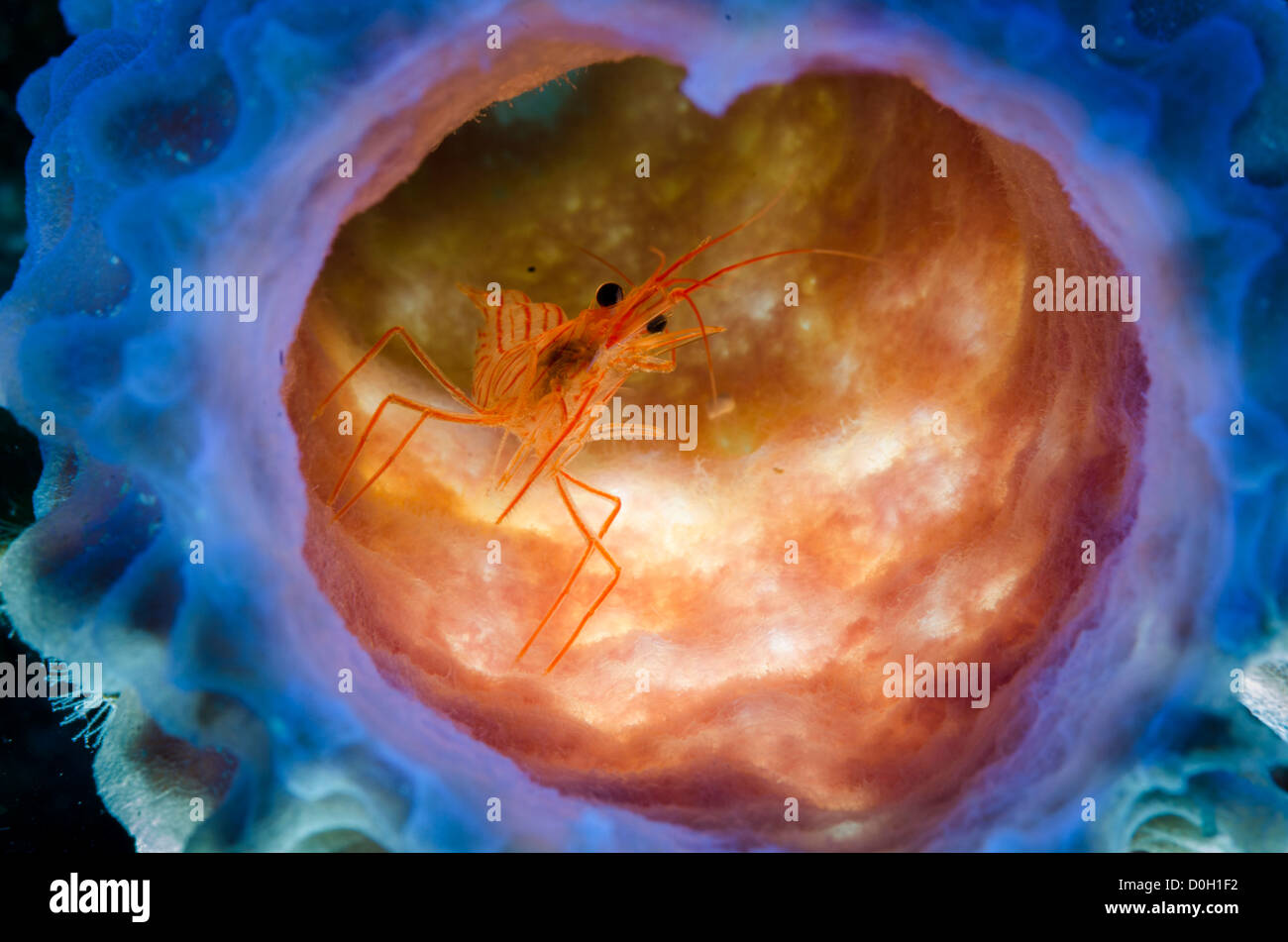
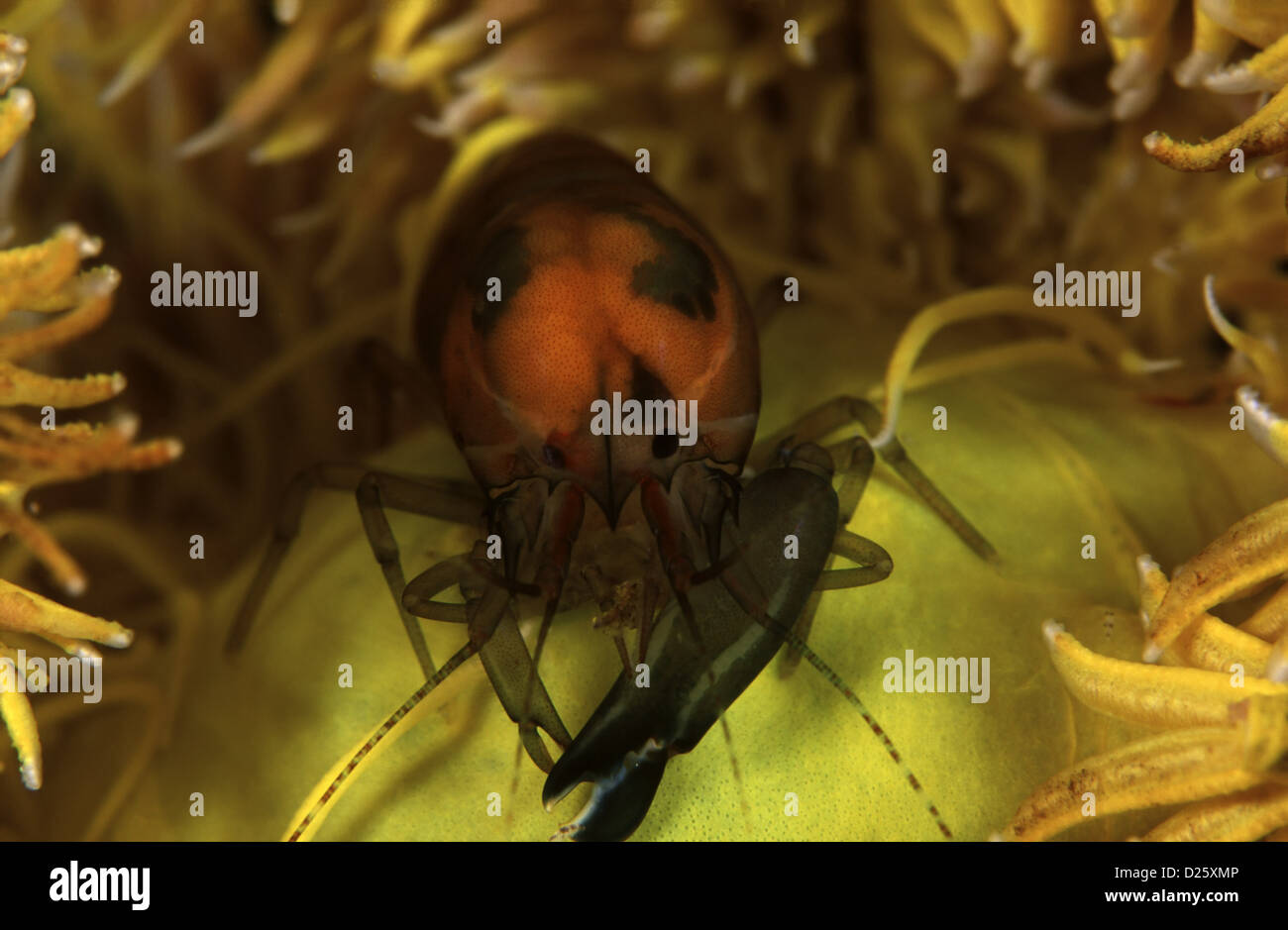
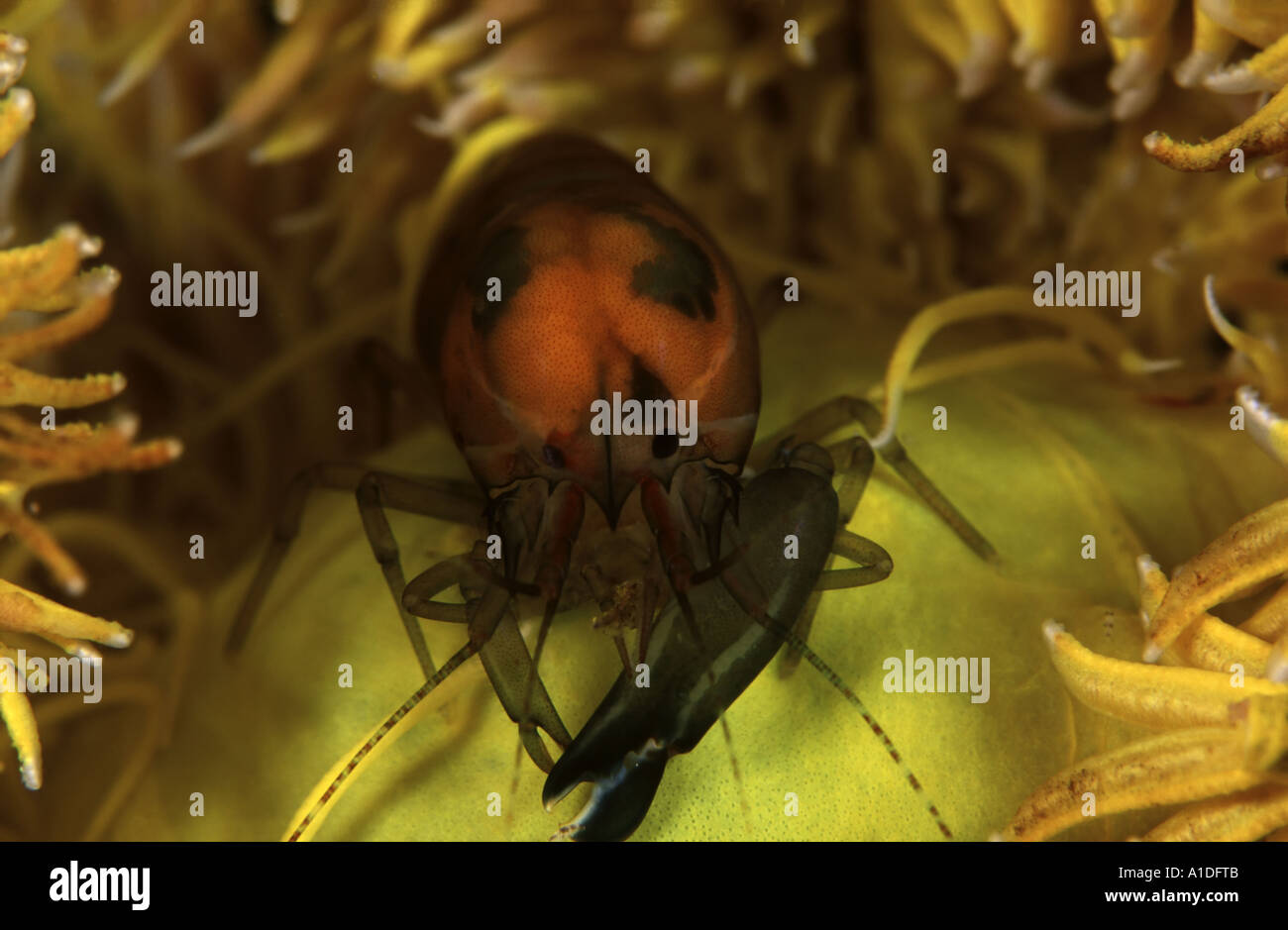
Synalpheus Stock Photos and Images

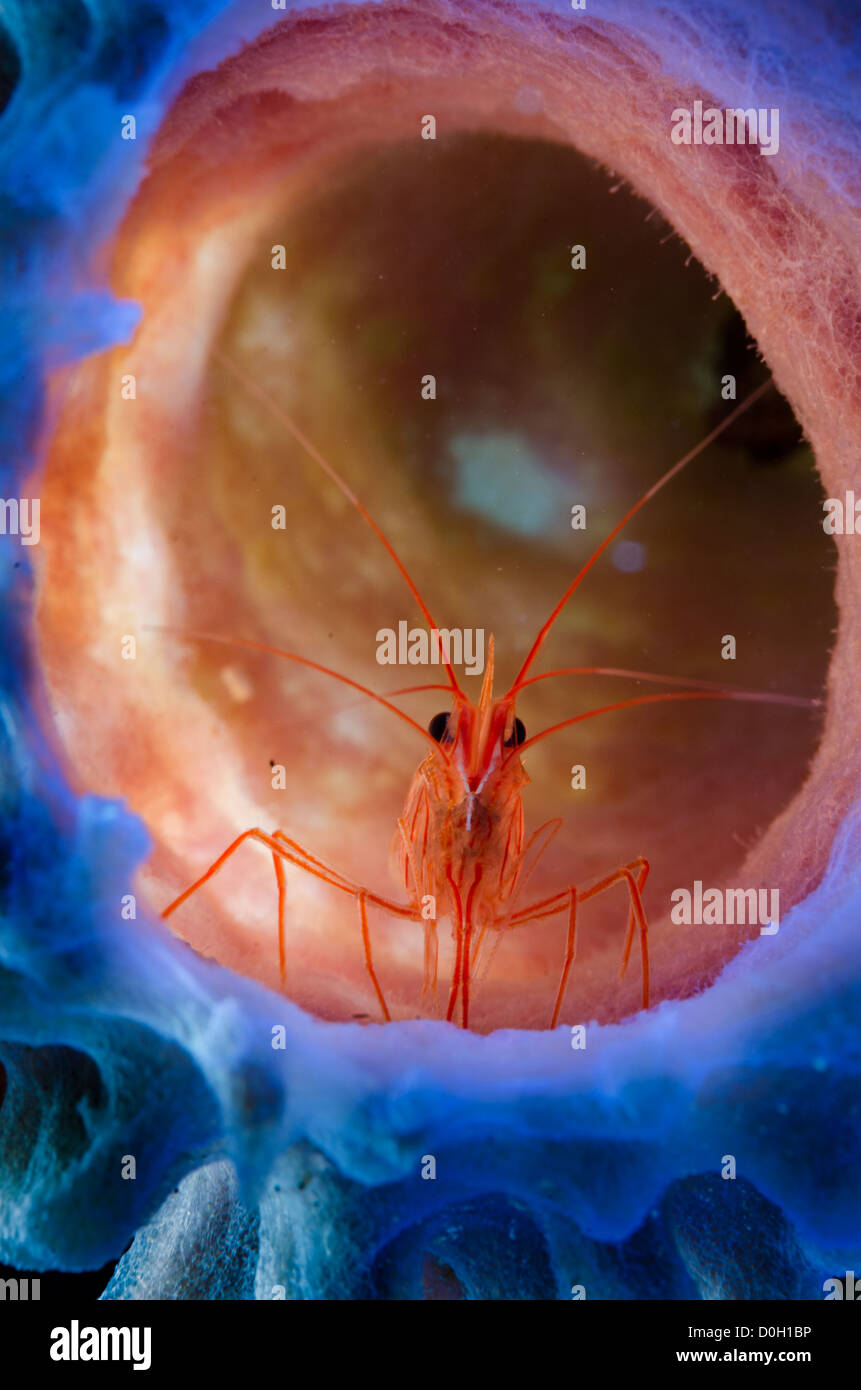
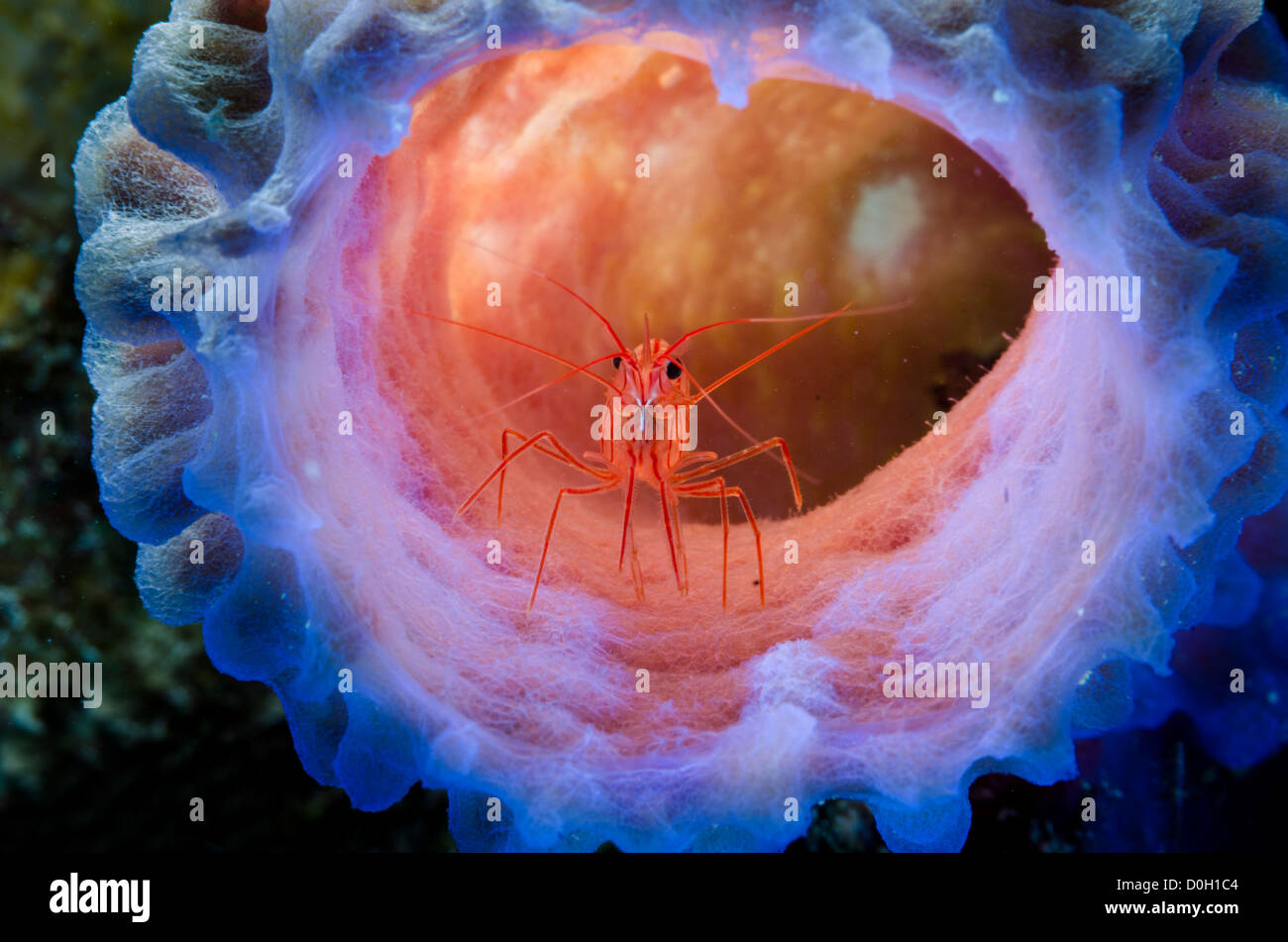
RMBJ5Y65–Stimpson’s Snapping Shrimps, Synalpheus stimpsoni, living underneath a crinoid, or featherstar.

RMHT5DN9–Crinoid Snapping Shrimps, Synalpheus stimpsoni, male and female living underneath a crinoid, or featherstar. Previously Synalpheus striatus.

RMR27DNR–Carinate snapping shrimp (Synalpheus carinatus) with eggs on a feather star, Walindi, Papua New Guinea

RM2FNXXAY–Crinoid Snapping Shrimp, Synalpheus stimpsoni, living underneath a crinoid, or featherstar. Previously Synalpheus striatus.Tulamben, Bali, Indonesia.

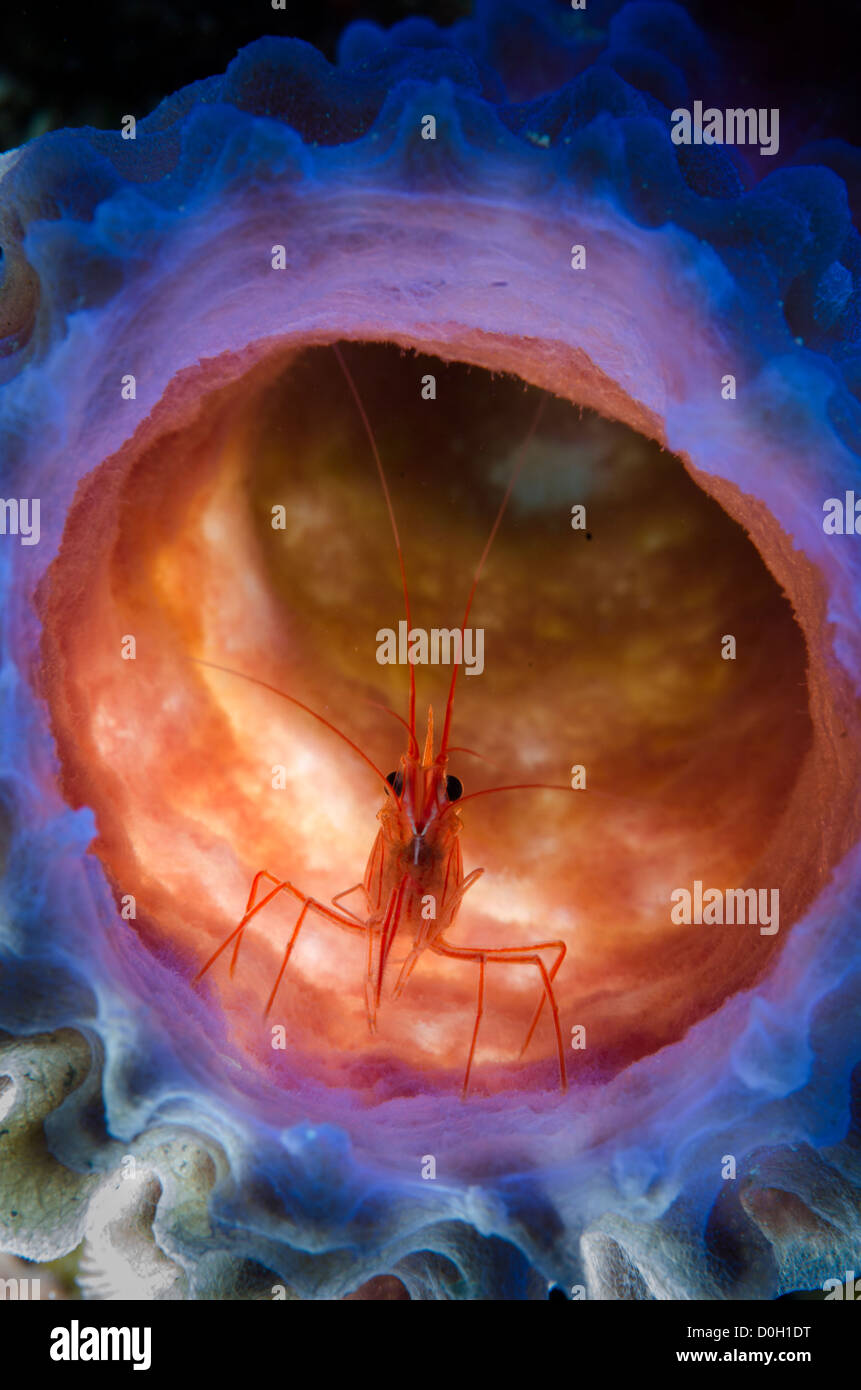
RMR27DM6–Stimpson's Snapping Shrimp (Synalpheus stimpsoni), inhabits only the central disc of crinoids (Comanthus sp.), Halmahera, Moluccas Sea, Indonesia

RM2B1HAC5–Crinoid Snapping Shrimp, Synalpheus stimpsoni, living underneath a crinoid, or featherstar. Previously Synalpheus striatus.Tulamben, Bali, Indonesia.

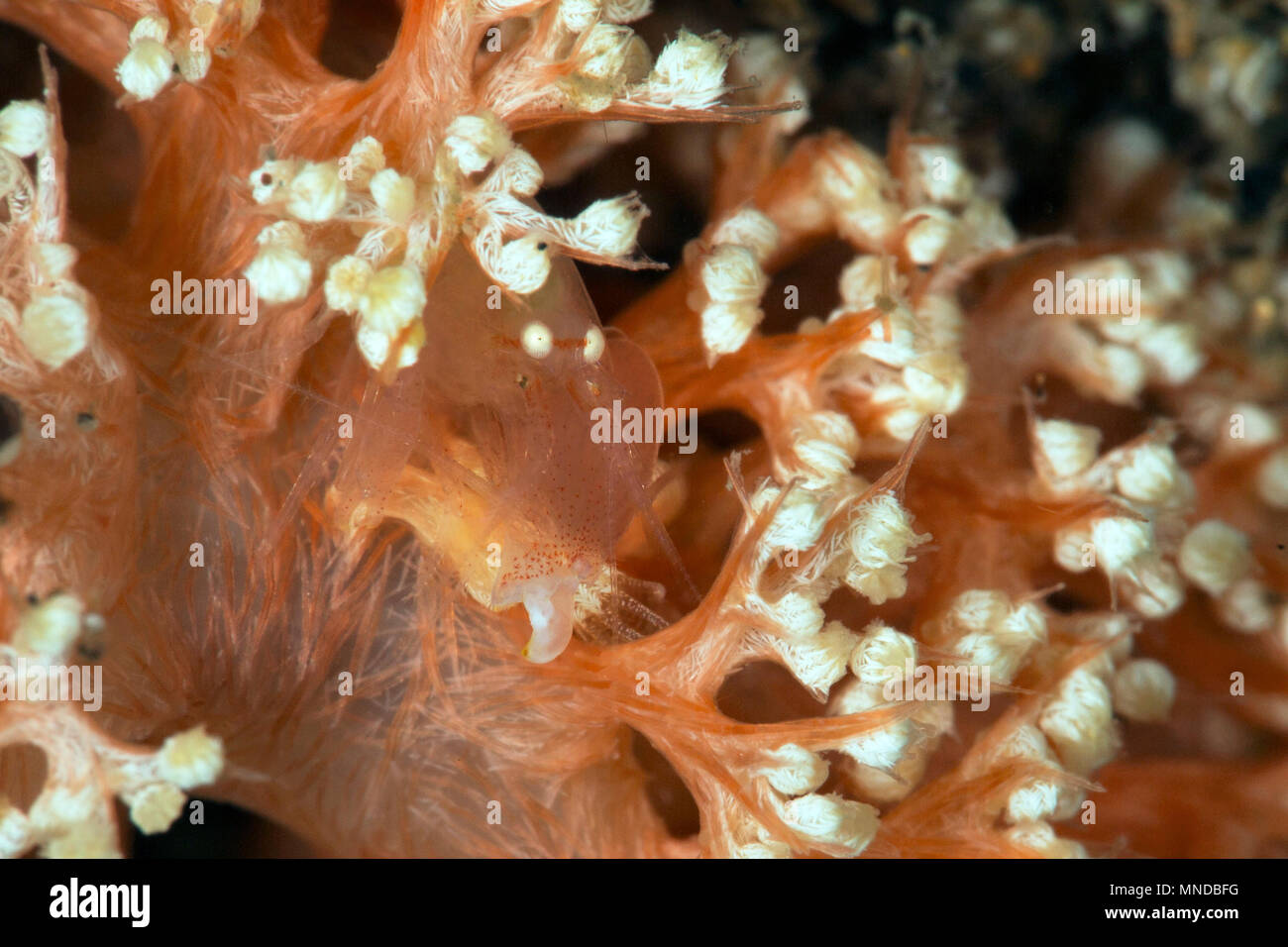
RM2PH43Y9–Sponge-dwelling Snapping Shrimp (Synalpheus sp.) adult, Lembeh Straits, Sulawesi, Sunda Islands, Indonesia

RM2D94ENP–Stimpson’s Snapping Shrimps, Synalpheus stimpsoni, living underneath a crinoid, or featherstar. Previously Synalpheus striatus. Uepi, Solomon Islands.

RM2R11K30–Black Crinoid Shrimp in feather star, Cabilao, Philippines, Philippine Sea, Asia (Synalpheus spec.)

RFEDJ8JD–Stimpson's Snapping Shrimp or crinoid snapping shrimp (synalpheus stimpsoni) Bohol Sea, Philippines, Southeast Asia

RM2PH43X9–Snapping Shrimp (Synalpheus sp.) adult female, with eggs, on reef at night Seraya, Bali, Lesser Sunda Islands, Indonesia

RMT3PBFK–De Man's Snapping Shrimp, Synalpheus demani, Alpheidae, Anilao, Batangas, Philippines, Philippine Sea, Indo-pacific Ocean, Asia

RMMARAY3–Snapping Prawn, Snapping Shrimp, Pistol Shrimp (Synalpheus stimpsoni) in the centre of a Crionoid..

RMR5BP61–Snapping Shrimp, Synalpheus cf demani, on Crinoid, Class Crinoidea, Pyramids dive site, Amed, east Bali, Indonesia, Indian Ocean

RM2C5RFKD–Striped Snapping Shrimp, Synalpheus striatus, on Crinoid, Comatulida Order, Pohon Miring dive site, Banda Besar Island, Banda Islands, Indonesia

RFPJPN4H–Snapping shrimp (Synalpheus neomeris) with eggs. Picture was taken in Lembeh strait, Indonesia

RM2AYYEX0–Modest snapping shrimp, Synalpheus modestus, Lembeh Strait, North Sulawesi, Indonesia, Pacific

RFPJPNX6–Modest Snapping Shrimp (Synalpheus modestus). Picture was taken in Lembeh strait, Indonesia

RMG9X221–Black Crinoid Shrimp in feather star, Cabilao, Philippines, Philippine Sea, Asia / (Synalpheus spec.)

RFDGH1KF–Striped Snapping Shrimp (Synalpheus striatus), mated pair on a crinoid in the Lembeh Strait off North Sulawesi, Indonesia.

RMDFWYWF–Striped snapping shrimp (Synalpheus striatus) on a yellow crinoid, Bunaken National Park, Sulawesi, Indonesia.
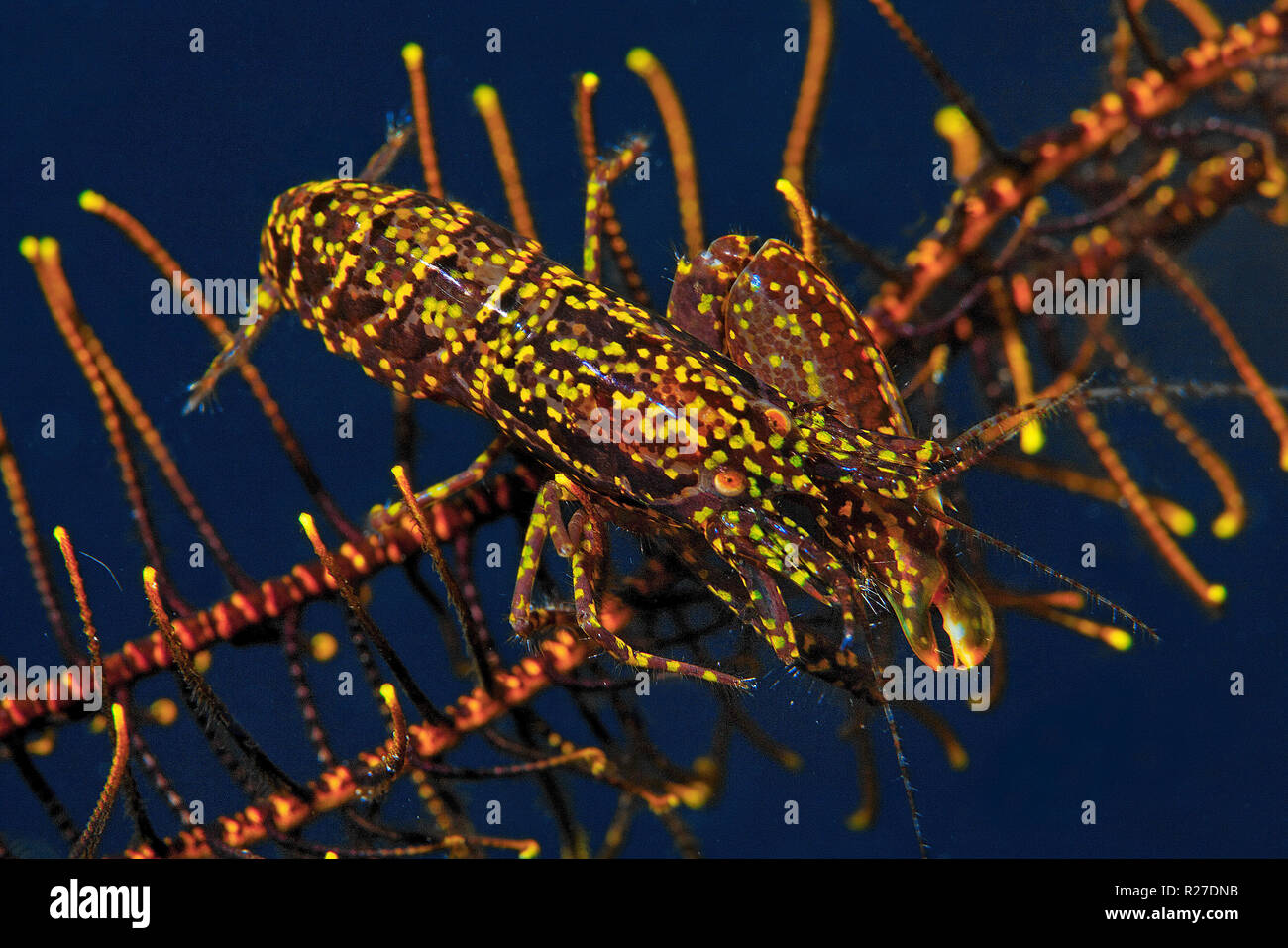
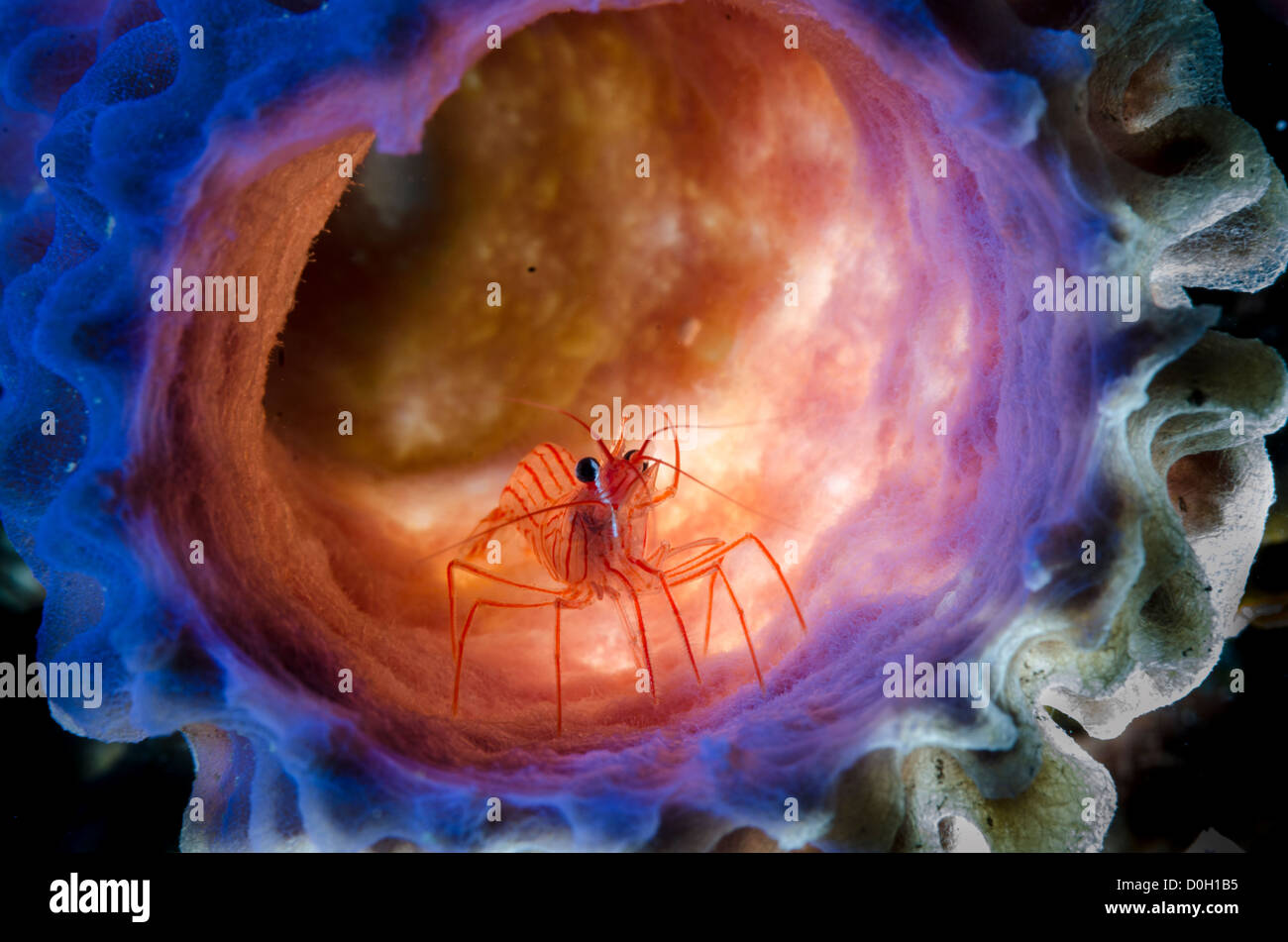
RMR27DNB–Stimpson's Snapping Shrimp (Synalpheus stimpsoni), inhabits only the central disc of crinoids (Comanthus sp.), Halmahera, Moluccas Sea, Indonesia

RF2AR9A39–Stimpson's Snapping Shrimp, Synalpheus stimpsoni on Crinoid, Crinoidea class, Nudi Retreat dive site, Lembeh Straits, Sulawesi, Indonesia, Pacific Oce

RMW7YF6H–Stimpson's snapping shrimp (Synalpheus stimpsoni) pair, female with eggs visible through transparent exoskeleton on host Crinoid (Comaster spp.) Lembeh Strait, North Sulawesi, Indonesia.

RMMFG2H2–Snapping shrimp, Synalpheus stimpsoni inside a crinoid or feather star, Halmahera, Moluccas Sea, Indonesia, Pacific Ocean

RM2PH43P4–Deman's Snapping Shrimp (Synalpheus demani) adult female, with eggs, on sand at reef, Seraya, Bali, Lesser Sunda Islands, Indonesia

RMW7X88F–Two Stimpson's snapping shrimps (Synalpheus stimpsoni) in a crinoid / feather star, Manado, Indonesia. Sulawesi Sea.

RMT3PBFT–De Man's Snapping Shrimp, Synalpheus demani, Alpheidae, Anilao, Batangas, Philippines, Philippine Sea, Indo-pacific Ocean, Asia

RMMFFJE4–PISTOL SHRIMP or CRACKER SHRIMP Synalpheus sp. Stuns its planktonic prey using long claw to generate shock wave

RM2C5RFNC–Recently moulted Striped Snapping Shrimp, Synalpheus striatus, on Crinoid, Comatulida Order, Pohon Miring dive site, Banda Besar Island, Banda Islands

RM2AYYEY8–Modest snapping shrimp, Synalpheus modestus, Lembeh Strait, North Sulawesi, Indonesia, Pacific

RFPKDB3D–Modest Snapping Shrimp (Synalpheus modestus). Picture was taken in Lembeh strait, Indonesia

RM2RRT8H4–Snapping Shrimp, Synalpheus sp, Bulakan Slope dive site, Seraya, Karangasem, Bali, Indonesia

RM2RETM9H–Snapping Shrimp, Synalpheus sp, on coral, Saonex Pier, Dampier Strait, Raja Ampat, West Papua, Indonesia

RMR27DME–Stimpson's Snapping Shrimp (Synalpheus stimpsoni), inhabits only the central disc of crinoids (Comanthus sp.), Halmahera, Moluccas Sea, Indonesia

RM2RG0NB2–Dark Knee Hermit Crab, Dardanus lagopodes, in shell with Snapping Shrimp, Synalpheus sp, Lombok Fields dive site, Seraya, Bali, Indonesia

RF2AR9A32–Stimpson's Snapping Shrimp, Synalpheus stimpsoni on Crinoid, Crinoidea class, Nudi Retreat dive site, Lembeh Straits, Sulawesi, Indonesia, Pacific Oce

RME292EK–Sponge-dwelling Snapping Shrimp (Synalpheus sp.) adult, Lembeh Straits, Sulawesi, Sunda Islands, Indonesia, January

RF2N4KYF1–Spearing Mantis Shrimp, Lysiosquillina sp, in hole with Snapping Shrimp, Synalpheus sp, Melasti dive site, Seraya, Karangasem, Bali, Indonesia, Indian

RM2PH43R0–Deman's Snapping Shrimp, Deman's Snapping Shrimp, Other animals, Crustaceans, Animals, Deman's Snapping Shrimp (Synalpheus demani) adult, on reef at

RFWXYEKY–Striped Snapping Shrimp, Synalpheus striatus, Sebayor Kecil dive site, between Komodo and Flores Islands, Komodo National Park, Indonesia, Indian Ocea

RMT3PBFP–De Man's Snapping Shrimp, Synalpheus demani, Alpheidae, Anilao, Batangas, Philippines, Philippine Sea, Indo-pacific Ocean, Asia

RFR6PY39–Stimpson's Snapping Shrimp or crinoid snapping shrimp (synalpheus stimpsoni) Bohol Sea, Philippines, Southeast Asia

RM2AM555C–Report on the scientific results of the voyage of H.M.SChallenger during the years 1873-76 : under the command of Captain George SNares, R.N., F.R.Sand Captain Frank Turle Thomson, R.N. . C.S.B adi SYNALPHEUS FALCATUS I PLATE CIV. PLATE CIV. Latreutes ensiferus (p. 583). Fig 1. Lateral view; enlarged six times. Id. Mandible. le. First siagnopod. If. Second siagnopod. (j. Third siagnopod. h. First gnathopod. i. Second gnathopod. Ik. Fiist pereiopod. 1/. Second pereiopod. lm. Third pereiopod ; distal joints, l^. Second pleopod, with ova attached. Platybema rugosum (p. 570).2. Lateral view ; e
![. Die Decapodenfauna der Adria : Versuch einer Monographie . Fig. 27. Synalpheus laevimanus (Heller). [Nach Contiere.] a) Stirnrand und Antennen von oben, Q. b) Große Schere des J. c) 3. Pereiopode. d) Telson. f) 2. Pereiopode. (Alle Figuren vergrößert.) Kritische Bemerkung zur Synonymie der Art: Heller (op. cit, 1863) ver- mutete, daß die von Costa (1838 in: Fauna del Eegno di Napoli) als Cryptophthalmus costa und Crypt. ventricosus beschriebenen und abgebildeten (Taf. 7, Fig.2A, B und Fig. 3 A, B) Alpheusarten mit dieser Form identisch seien, vielleicht nur die beiden Geschlechter von A. lae Stock Photo . Die Decapodenfauna der Adria : Versuch einer Monographie . Fig. 27. Synalpheus laevimanus (Heller). [Nach Contiere.] a) Stirnrand und Antennen von oben, Q. b) Große Schere des J. c) 3. Pereiopode. d) Telson. f) 2. Pereiopode. (Alle Figuren vergrößert.) Kritische Bemerkung zur Synonymie der Art: Heller (op. cit, 1863) ver- mutete, daß die von Costa (1838 in: Fauna del Eegno di Napoli) als Cryptophthalmus costa und Crypt. ventricosus beschriebenen und abgebildeten (Taf. 7, Fig.2A, B und Fig. 3 A, B) Alpheusarten mit dieser Form identisch seien, vielleicht nur die beiden Geschlechter von A. lae Stock Photo](https://c8.alamy.com/comp/MEE80G/die-decapodenfauna-der-adria-versuch-einer-monographie-fig-27-synalpheus-laevimanus-heller-nach-contiere-a-stirnrand-und-antennen-von-oben-q-b-groe-schere-des-j-c-3-pereiopode-d-telson-f-2-pereiopode-alle-figuren-vergrert-kritische-bemerkung-zur-synonymie-der-art-heller-op-cit-1863-ver-mutete-da-die-von-costa-1838-in-fauna-del-eegno-di-napoli-als-cryptophthalmus-costa-und-crypt-ventricosus-beschriebenen-und-abgebildeten-taf-7-fig2a-b-und-fig-3-a-b-alpheusarten-mit-dieser-form-identisch-seien-vielleicht-nur-die-beiden-geschlechter-von-a-lae-MEE80G.jpg)
RMMEE80G–. Die Decapodenfauna der Adria : Versuch einer Monographie . Fig. 27. Synalpheus laevimanus (Heller). [Nach Contiere.] a) Stirnrand und Antennen von oben, Q. b) Große Schere des J. c) 3. Pereiopode. d) Telson. f) 2. Pereiopode. (Alle Figuren vergrößert.) Kritische Bemerkung zur Synonymie der Art: Heller (op. cit, 1863) ver- mutete, daß die von Costa (1838 in: Fauna del Eegno di Napoli) als Cryptophthalmus costa und Crypt. ventricosus beschriebenen und abgebildeten (Taf. 7, Fig.2A, B und Fig. 3 A, B) Alpheusarten mit dieser Form identisch seien, vielleicht nur die beiden Geschlechter von A. lae

RM2AYAJA1–Soft coral snapping shrimp, Synalpheus neomeris, Lembeh Strait, North Sulawesi, Indonesia, Pacific

RFPJPNXG–Modest Snapping Shrimp (Synalpheus modestus). Picture was taken in Lembeh strait, Indonesia

RMR27DMW–Stimpson's Snapping Shrimp (Synalpheus stimpsoni), inhabits only the central disc of crinoids (Comanthus sp.), Halmahera, Moluccas Sea, Indonesia

RMCNN6NP–Deman's Snapping Shrimp Synalpheus demani adult reef night Uhak Reef Wetar Island Barat Daya Islands Lesser Sunda Islands

RM2CBY2BE–Juvanile red-coral pistol shrimp, Synalpheus charon, Alpheidae, Anilao, Batangas, Philippines, Philippine Sea, Indo-pacific Ocean, Asia
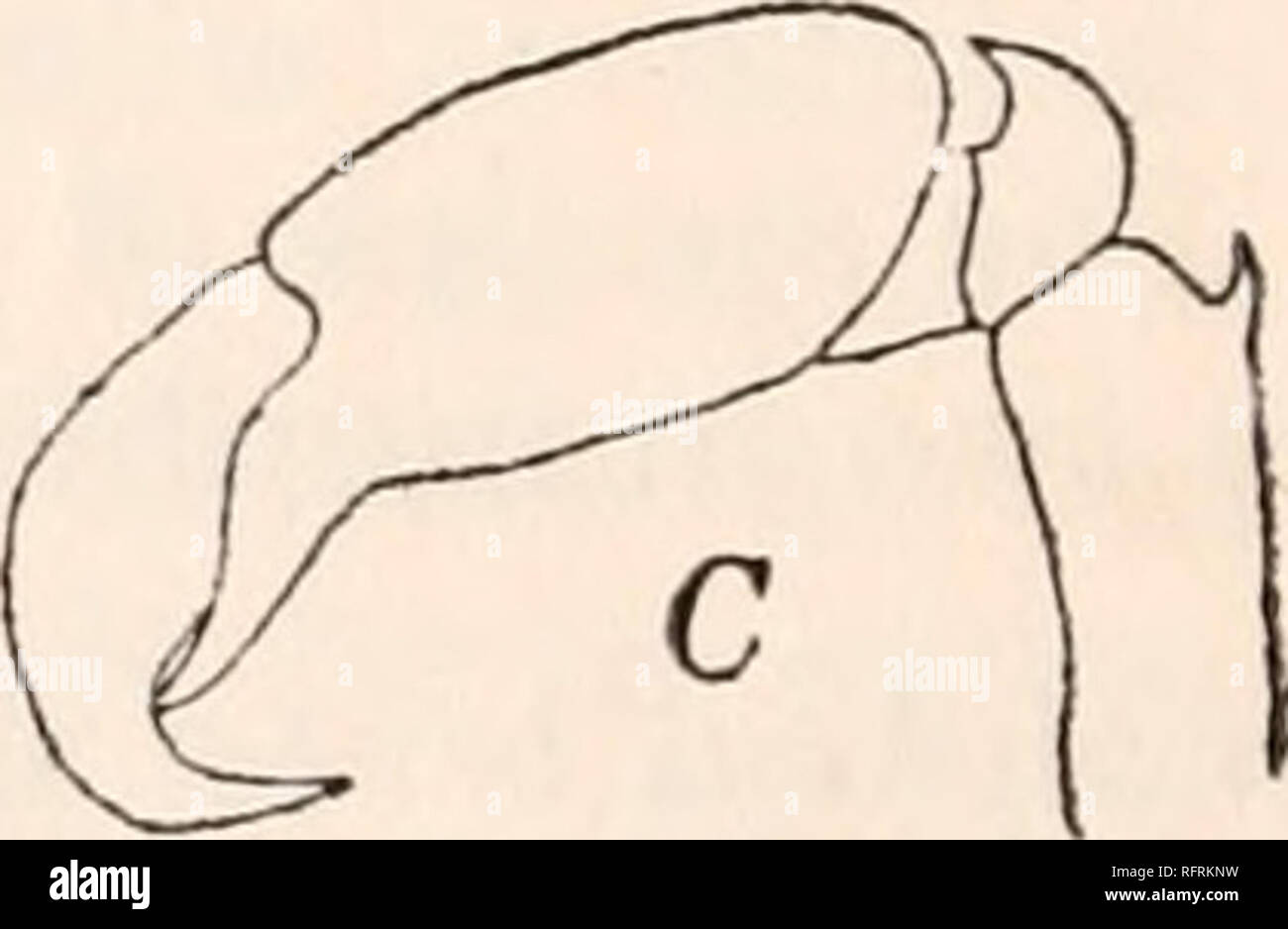
RMRFRKNW–. Carnegie Institution of Washington publication. . FIG. 1.—Chelse of Synalpheus. 8. brucei: A, large chela, X 4; B, small chela, X 8. S. comatularum: C, small chela, X 8.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Carnegie Institution of Washington. Washington, Carnegie Institution of Washington

RMDGHH2E–Snapping Shrimp (Synalpheus sp.) adult female, with eggs, on reef at night Seraya, Bali, Lesser Sunda Islands, Indonesia, April
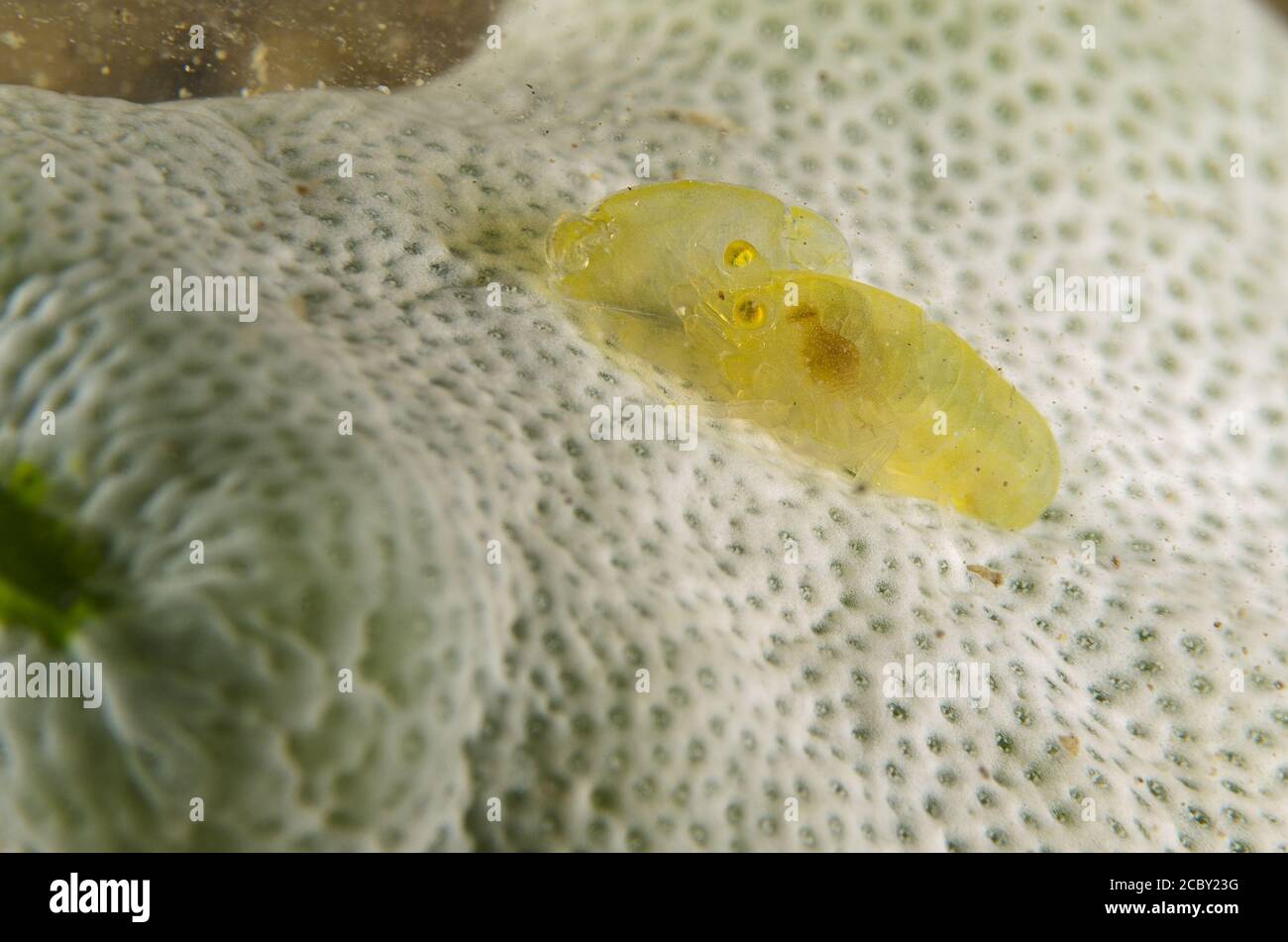
RM2CBY23G–Juvanile red-coral pistol shrimp, Synalpheus charon, Alpheidae, Anilao, Batangas, Philippines, Philippine Sea, Indo-pacific Ocean, Asia

RMRFRGCP–. Carnegie Institution of Washington publication. . 1. Synalpheus brooks! Coutii-re. 2. Chela of same species to show different colorations. 3. The Pontoniid commensal with the above. B.Meisellith 4. Cradactis variabilis Hargitt. 5. Cradactis variabilis Harftitl. 6. The same, showing a third variation.. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Carnegie Institution of Washington. Washington, Carnegie Institution of Wa

RMDGHH1P–Deman's Snapping Shrimp (Synalpheus demani) adult female with eggs on sand at reef Seraya Bali Lesser Sunda Islands Indonesia

RM2CBY23J–Juvanile red-coral pistol shrimp, Synalpheus charon, Alpheidae, Anilao, Batangas, Philippines, Philippine Sea, Indo-pacific Ocean, Asia
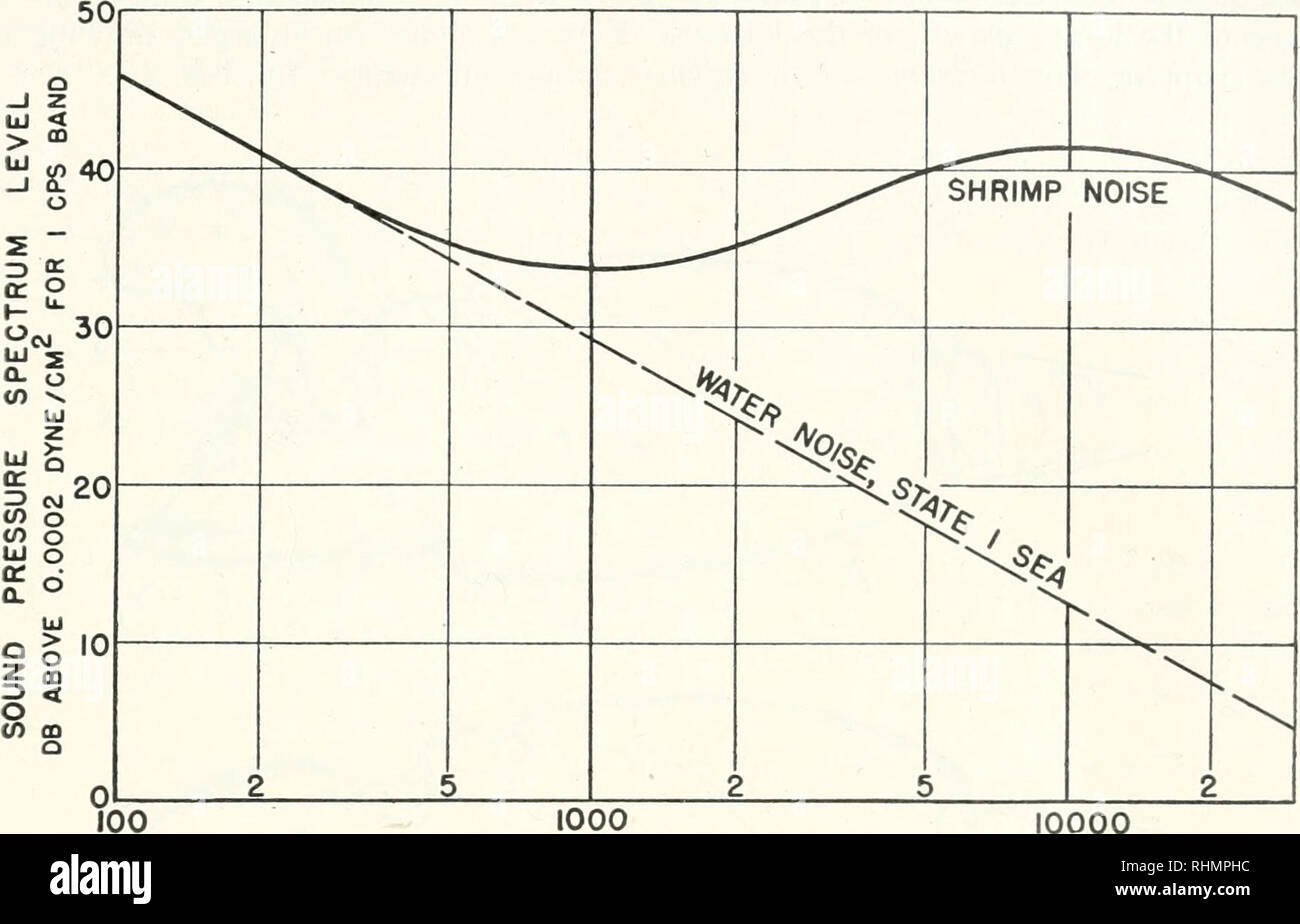
RMRHMPHC–. The Biological bulletin. Biology; Zoology; Biology; Marine Biology. SNAPPING SHRIMP AND UNDERWATER NOISE 127 ever, they are very unlike the common shrimp. The snapping shrimp family Crangonidae (Alpheidae, see Rathbun, 1904) comprises about 27 genera and numerous species. Of these only the species of two genera, viz., Crangon (also called Alpheus by many authors) and Synalpheus, are capable of vigorous snap- ping. In the literature there are recorded about 215 species of Crangon and 150 species of Synalpheus. Two species of these genera are shown in Figures 1 and 2.. 1000 10000 FREQUENCY IN
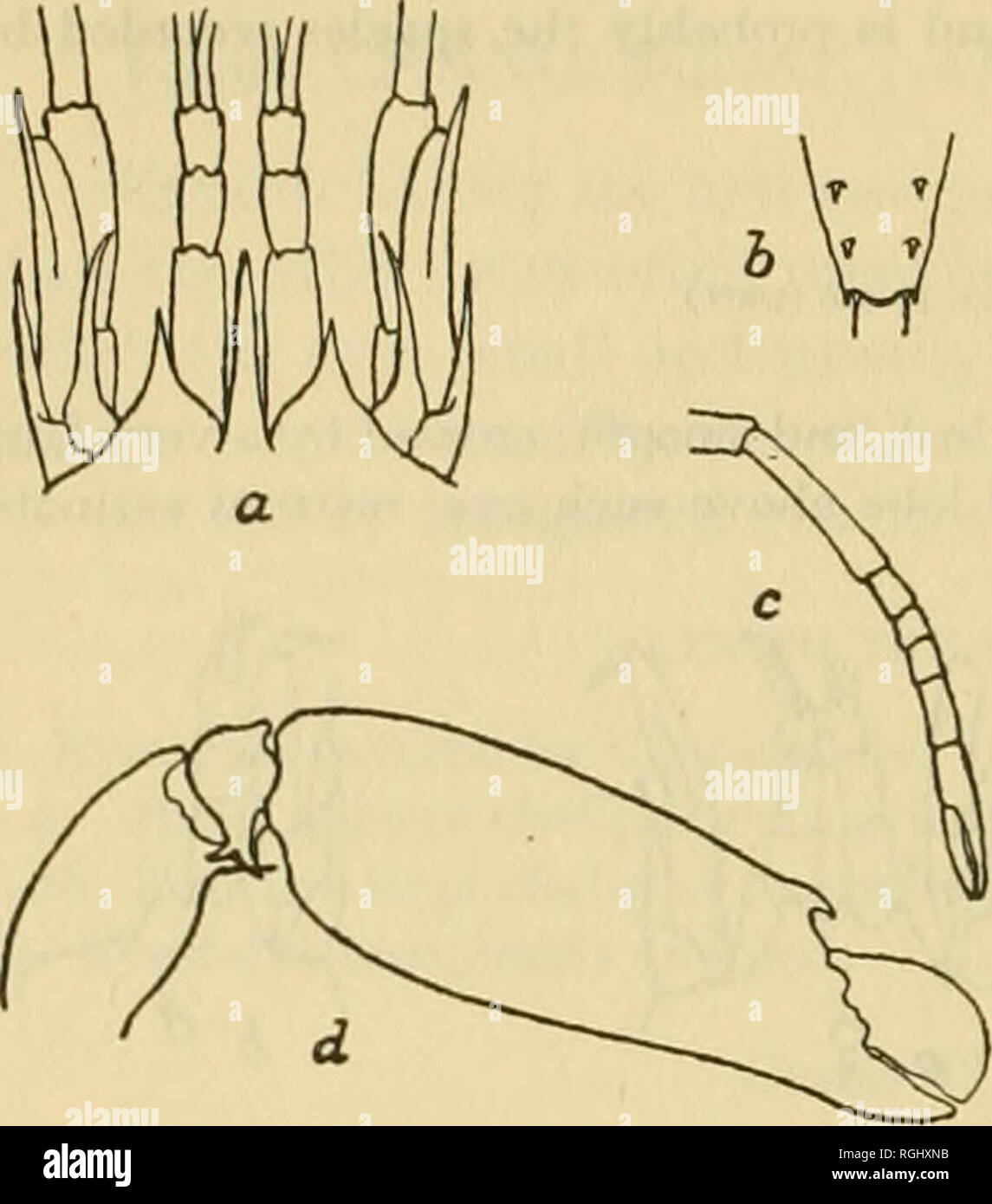
RMRGHXNB–. Bulletin of the Bureau of Fisheries. Fisheries; Fish culture. 384 BULLETIN OF THE BUREAU OP FISHERIES.. Synalpheus townsendi Couti^e. PL xxvi, fig. i. Synalpheus townsendi Coutifere, 1909, p. 32. Similar in form to S. longicarpus but with a much slenderer rostrum which considerably exceeds the supra-orbital lobes and reaches slightly beyond the distal end of the basal article of the antennule. The sides of the telson are not as strongly convergent and are slightly produced into little angles at the distal end and the inner pair of spines are slender and about three times as long as the outer
RMRHK2XM–. The biology of marine animals. Marine animals; Physiology, Comparative. SENSORY ORGANS AND RECEPTION 341 Invertebrates. A few invertebrates are reported to be sensitive to sounds and water-borne vibrations. Underwater sounds evoke withdrawal (startle) reflexes in sabellid worms, e.g. Branchiomma. Some crustaceans produce sounds, e.g. Palinurus, Synalpheus, Uca (p. 405), and may have limited auditory ability. It is known that Mysis and Palaemonetes can hear underwater sounds. Bethe observed that they are more responsive to deep tones and that sensitivity is reduced after extirpation of the st
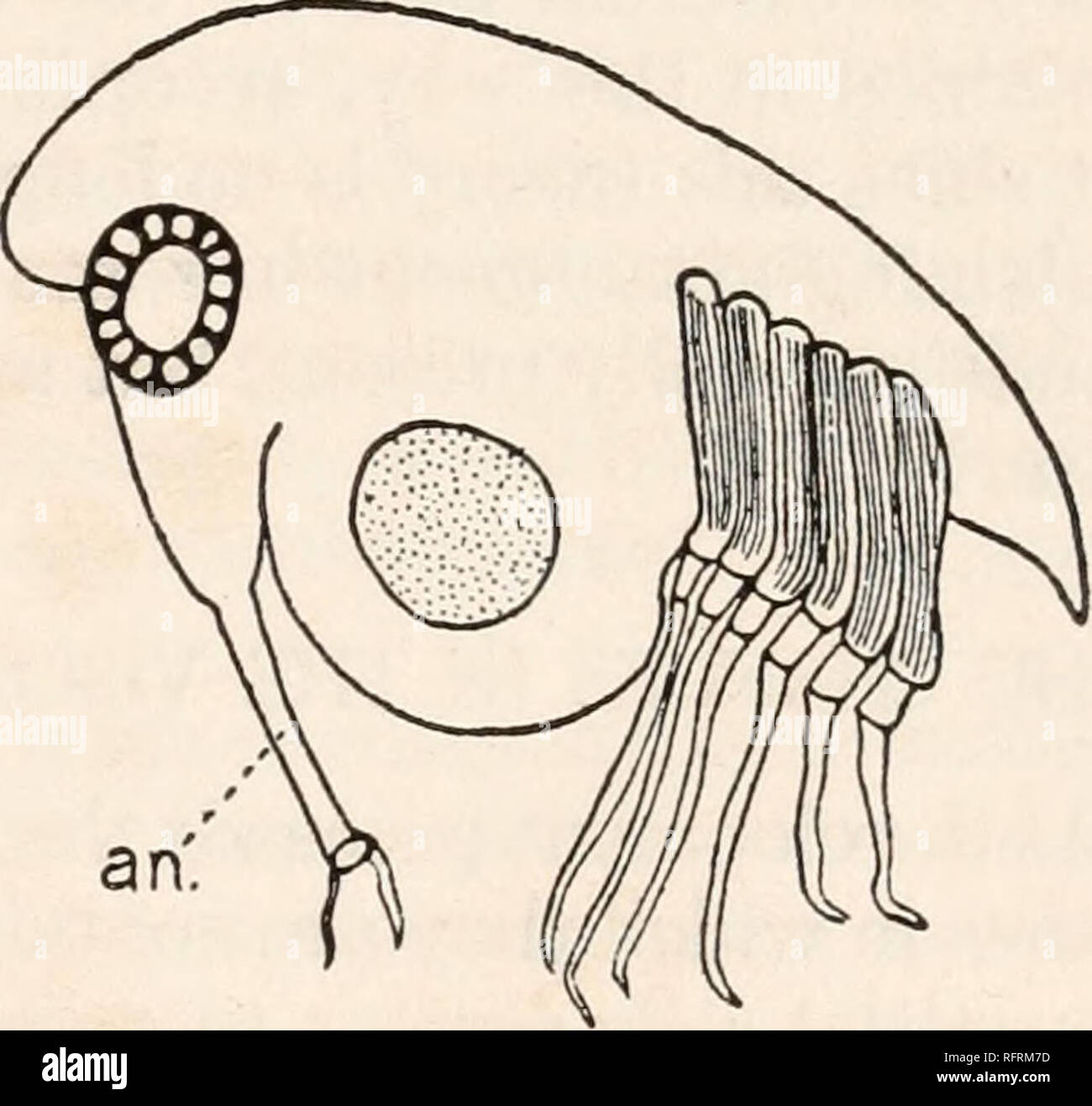
RMRFRM7D–. Carnegie Institution of Washington publication. . an. FIG. 10.—"Pro-Cypris" larva of Thompsonia from Thalamita in egg-membrane. X375. FIG. 11.—Cypris larva of Thompsonia from Synalpheus. The dotted area in the ventral part of the body contains reserve materials, an., antenna. X470. within the egg membrane. While, moreover, the egg of Sacculina is comparatively large, 600 to 800 fj. in diameter, that of Thompsonia measures only about 34 //. Segmentation commences unequally, a micromere and a macromere with all the yolk being formed. Both micromere and macromere take part in the seco
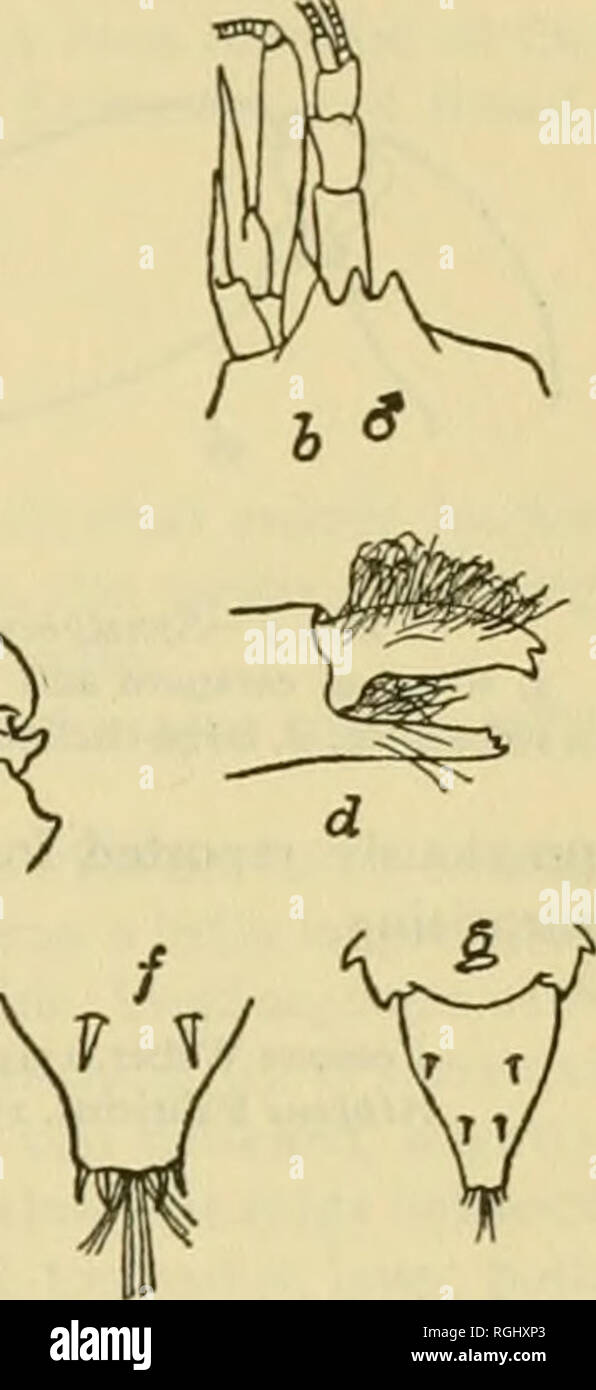
RMRGHXP3–. Bulletin of the Bureau of Fisheries. Fisheries; Fish culture. ^H^ Alpheus saulcyi var. longicarpus Herrick, in Brooks and Herrick, 1892, p. 383 (part). Synalpheus longicarpus Couti^re, 1909. p. 53. Carapace about two-thirds as long as abdomen, subcylindrical, and smooth; crossed by a very faint cervical groove; frontal border produced into a sharp-pointed lobe above each eye,- rostrum carinate, slender and slightly longer than supra-orbital lobes. Abdomen smooth, tapering; telson with strongly con- vergent, sinuous sides and truncate tip which bears four slender spines, upper surface with fo
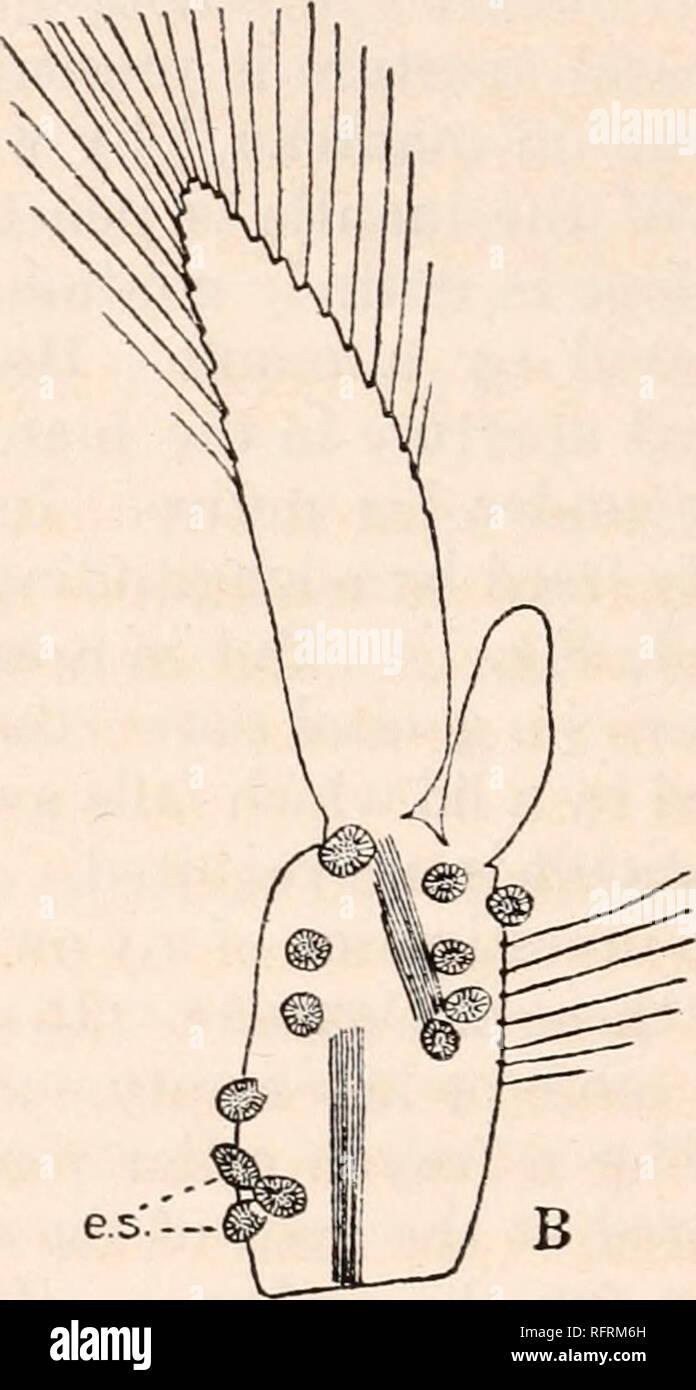
RMRFRM6H–. Carnegie Institution of Washington publication. . e.s— B FIG. 12.—First abdominal appendage (right hand side) of Synalpheus brucei bearing external sacs of Thompsonia. X28. A. Cast skin showing external sacs (e. s.) containing nearly mature larvae. Only two sacs are still present, the others having been accidentally detached, but the round black rings (ped.) indicate their position. B. The same appendage 3 days after moulting, showing the new crop of external sacs. number of pink bodies much smaller than any observed hitherto, but with the characteristic structure of the external sac, were f
![. Annals of the South African Museum. Annale van die Suid-Afrikaanse Museum. Natural history. Aim. S. Afr. Mus. Vol. XV. Crustacea Plate LXXXVII. Plate XXIII. prp prp.l.. -Del.T.R.R.Stebting. West,Newman lith. SYNALPHEUS AN I S OCHEIR. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. South African Museum; South African Museum. Annale van die Suid-Afrikaanse Museum. Cape Town [etc. , South African Museum, etc. ] Stock Photo . Annals of the South African Museum. Annale van die Suid-Afrikaanse Museum. Natural history. Aim. S. Afr. Mus. Vol. XV. Crustacea Plate LXXXVII. Plate XXIII. prp prp.l.. -Del.T.R.R.Stebting. West,Newman lith. SYNALPHEUS AN I S OCHEIR. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. South African Museum; South African Museum. Annale van die Suid-Afrikaanse Museum. Cape Town [etc. , South African Museum, etc. ] Stock Photo](https://c8.alamy.com/comp/RMJEJ2/annals-of-the-south-african-museum-annale-van-die-suid-afrikaanse-museum-natural-history-aim-s-afr-mus-vol-xv-crustacea-plate-lxxxvii-plate-xxiii-prp-prpl-deltrrstebting-westnewman-lith-synalpheus-an-i-s-ocheir-please-note-that-these-images-are-extracted-from-scanned-page-images-that-may-have-been-digitally-enhanced-for-readability-coloration-and-appearance-of-these-illustrations-may-not-perfectly-resemble-the-original-work-south-african-museum-south-african-museum-annale-van-die-suid-afrikaanse-museum-cape-town-etc-south-african-museum-etc-RMJEJ2.jpg)
RMRMJEJ2–. Annals of the South African Museum. Annale van die Suid-Afrikaanse Museum. Natural history. Aim. S. Afr. Mus. Vol. XV. Crustacea Plate LXXXVII. Plate XXIII. prp prp.l.. -Del.T.R.R.Stebting. West,Newman lith. SYNALPHEUS AN I S OCHEIR. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. South African Museum; South African Museum. Annale van die Suid-Afrikaanse Museum. Cape Town [etc. , South African Museum, etc. ]
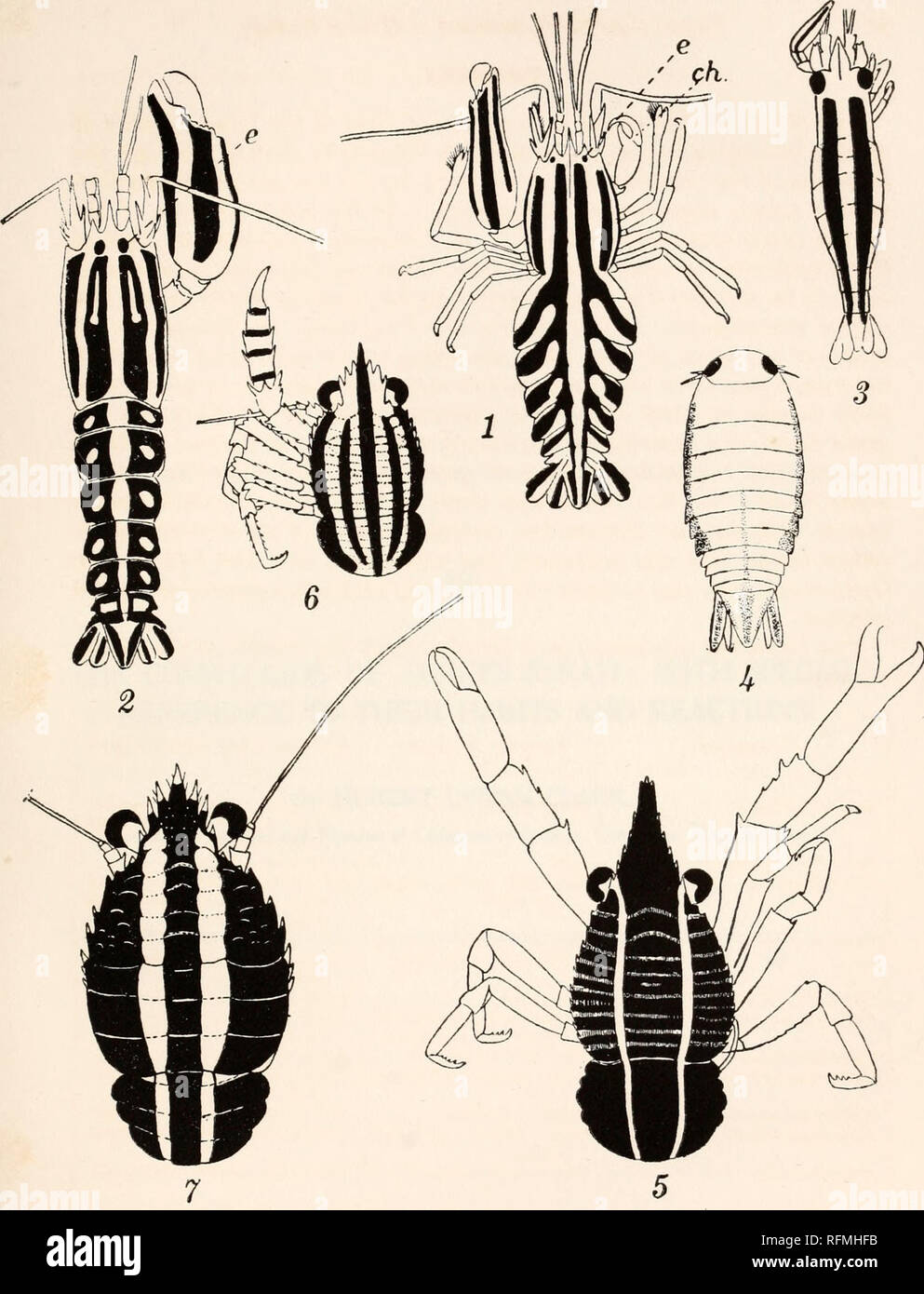
RMRFMHFB–. Carnegie Institution of Washington publication. F. A POTTS PLATE 1. FIG. 1 Synalpheus comatularum. X1.5. Fio.j[2. S. brucei: e, eye; ch., small chela with its incurved thumb. X3. Only a single example of S. comatularum and another of S. brucei is figured, so no idea of the variation in extent of pigmentation is shown, but only the type of arrangement. But both the type and variation are alike in the two species, that of S. brucei having rather deeper pigmentation, the bands being wider and the lateral unpigmented areas in the ab- dominal segments being completely enveloped by pigment. FIG. 3
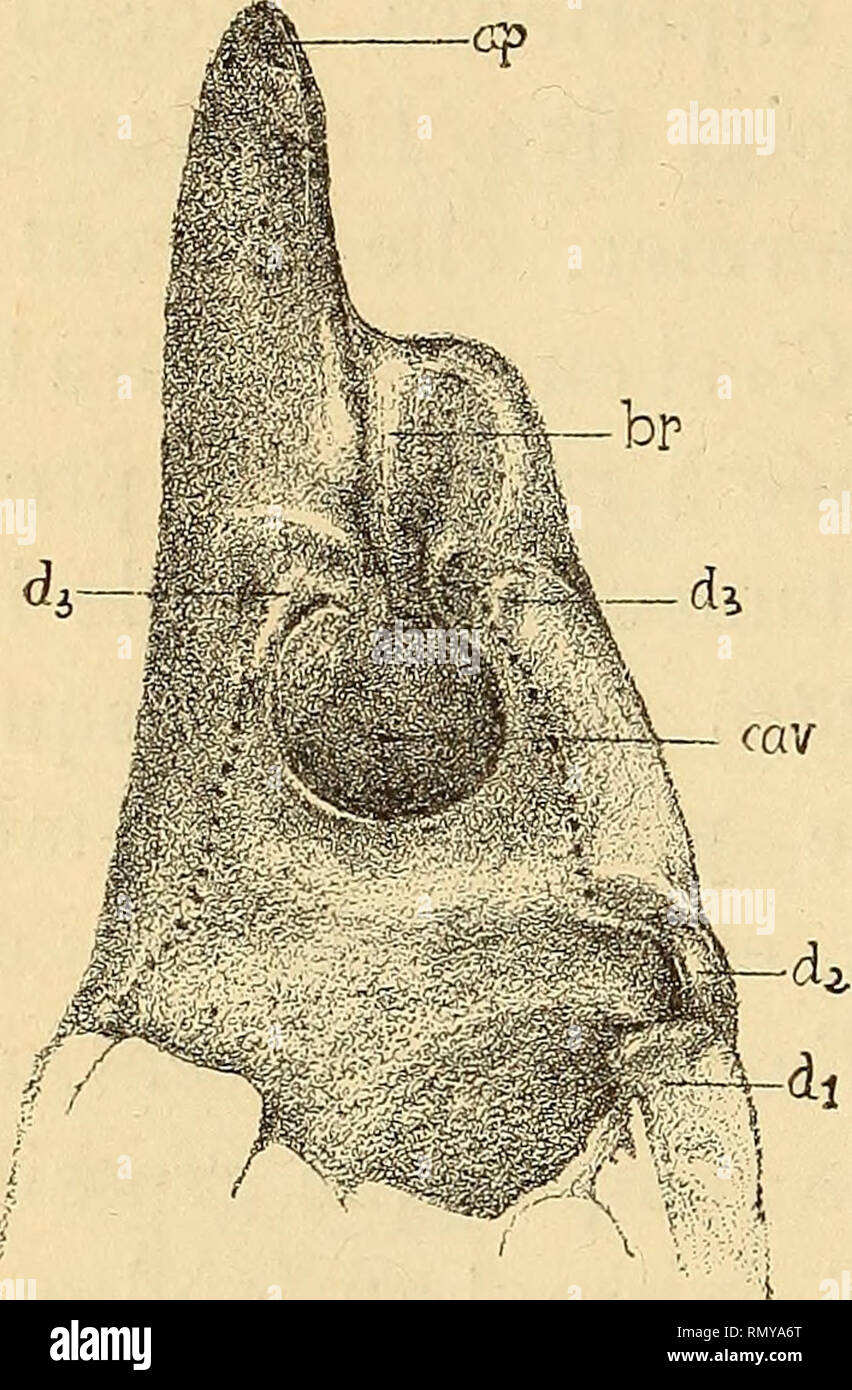
RMRMYA6T–. Annales des sciences naturelles. Zoology; Biology. 204 H. COUTlEltE. (supéro-interne) de la pince, est l'homologue de la dent située chez Alpheopsis en un point correspondant. Cette saillie possède chez Synalpheus un bord horizontal {^^. 249 et 250) parallèle au grand axe de la pince, et un bord ver- tical, l'un et l'autre bien marqués. Le bord vertical limite la brèche citée plus haut, puis, s'infléchissant un peu en. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations

RMRMJFGN–. Annals of the South African Museum = Annale van die Suid-Afrikaanse Museum. Natural history. Descriptive Catalogue of South African Decapod Crustacea. 737 of dorsolateral spinules, anterior pair at middle of length, 2 unequal spines in notch on either side of the gently convex apical margin. Length $ up to 26 mm., ova 2 mm. diam.. Fig. 139.—Synalpheus anisocheir Stebb. a, carapace, b, dorsal view of rostrum and bases of 1st and 2nd antennae, c, inner view of chela of 1st leg $, with tooth at end of hand as developed in some specimens, d, posterior view of dactyl and apex of 6th joint of 5th
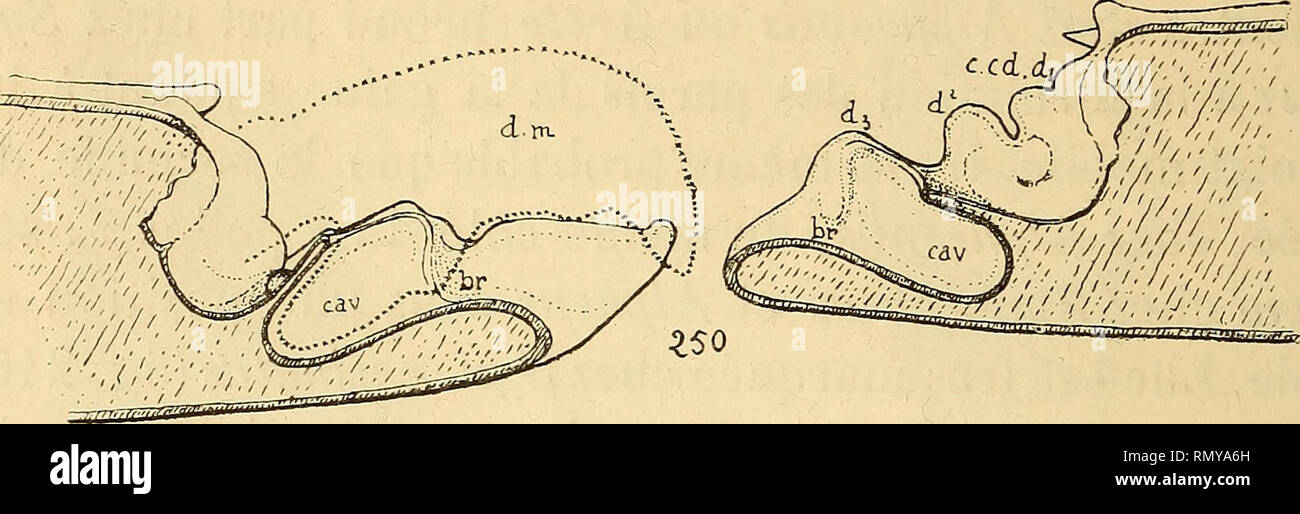
RMRMYA6H–. Annales des sciences naturelles. Zoology; Biology. 206 H. COUTIËRi:. QiQxSynalpheus au contraire, raccroissement des deux bords a été presque symétrique, réalisant ainsi une disposition nouvelle (fig. 249). Aucun autre Crustacé Décapode n'offre, à ce degré, Tengainement du doigt mobile par le « pollex » qui caracté- rise les Alphéidés supérieurs, Synalpheus^ Alpheus^ et jus- qu'à un certain point, Amphibetdeus. Je montrerai plus loin que le mécanisme de ce singulier appareil d'attaque et de. Please note that these images are extracted from scanned page images that may have been digitally en
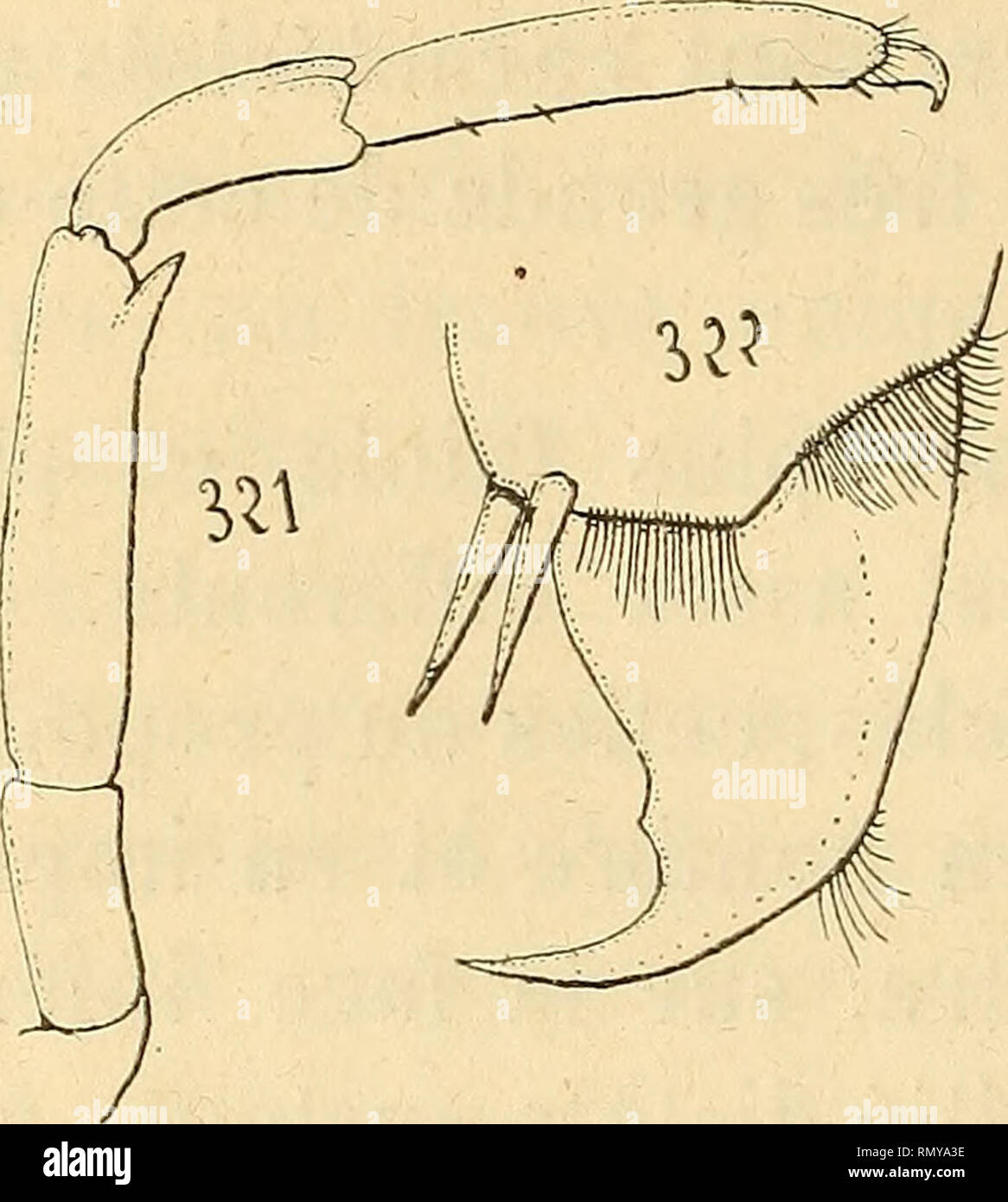
RMRMYA3E–. Annales des sciences naturelles. Zoology; Biology. 262 H. COUTIERi:. Synalpheus comatularum^ S. Stimpsoni, S. can?iatus. Les autres espèces de Synalpheus se montrent en général mieux pourvues et possèdent de 4 à 10 rangées de soies obliques (fig. 326). Chez Alpheus, enfin, les rangées de soies de la 5' paire ne. Please note that these images are extracted from scanned page images that may have been digitally enhanced for readability - coloration and appearance of these illustrations may not perfectly resemble the original work.. Milne-Edwards, H. (Henri), 1800-1885; Audouin, Jean Victor, 179
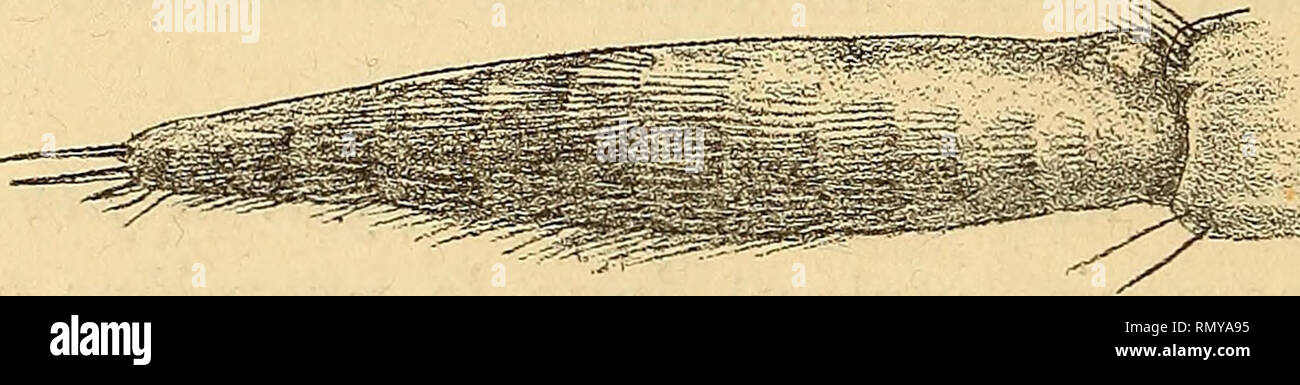
RMRMYA95–. Annales des sciences naturelles. Zoology; Biology. lo'l.. Fig. '201. Alplieus lœvis, Randall, maxillipède lU. — Fig. 202. Alhanas nitescens, Leach, maxillipède 111, article distal. lipèdes externes relativement court et inerme ; ce carac- tère se maintient sans changements chez la plupart des Alphéidés, mais il évolue chez quelques-uns d'entre eux {Automate, Synalpheus) dans un sens exactement compa- rable à celui qui caractérise les Hippolytidés. Dans cette dernière famille, l'allongement excessif et l'armature épineuse distale des rhaxillipèdes sont, du reste, manifestement des caractères
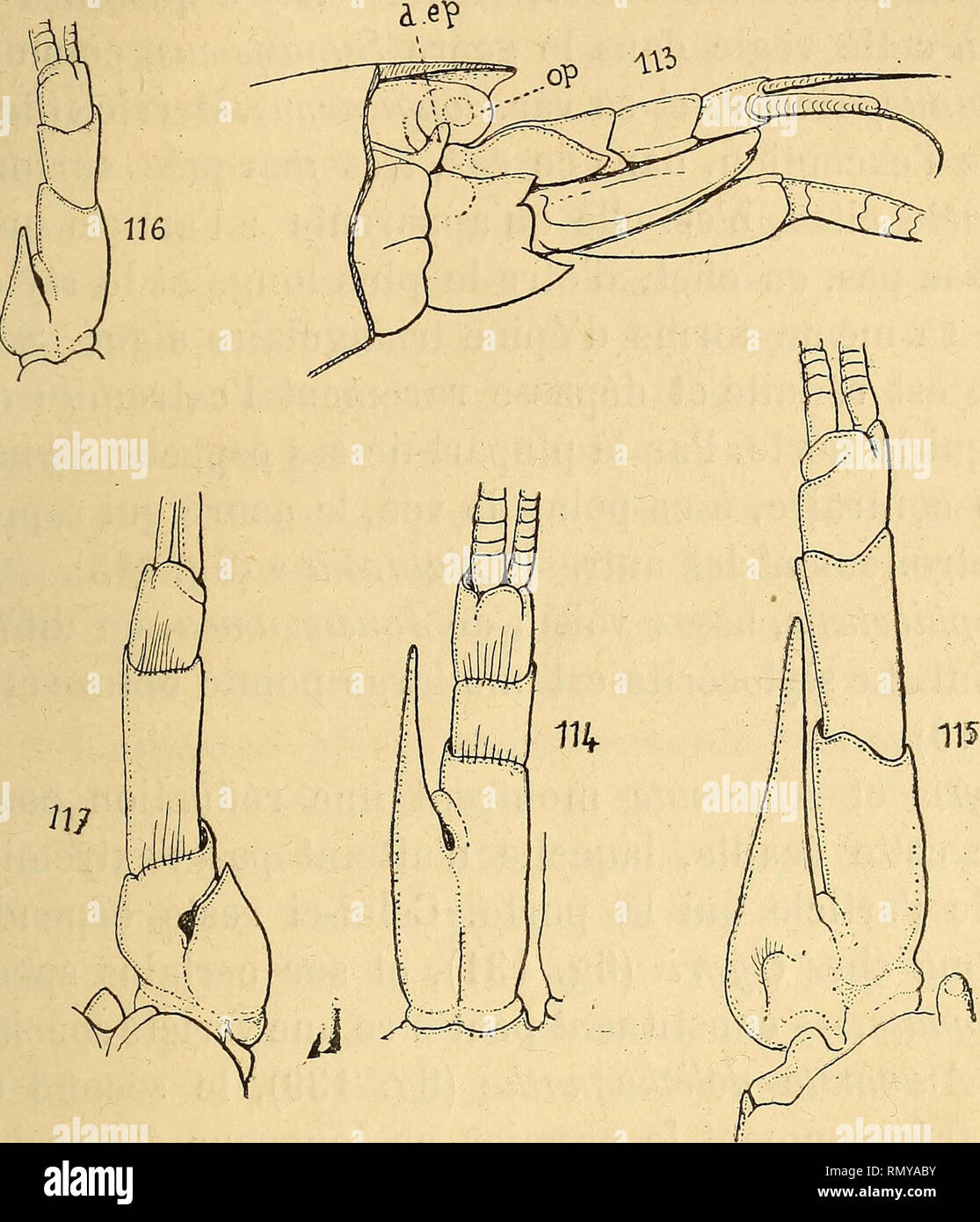
RMRMYABY–. Annales des sciences naturelles. Zoology; Biology. ALPHEID^, 12' terminée en pointe et très développée, constitue chez les <( Natantia » une protection de la région frontale qui manque rarement, ou qui, en cas d'absence, est toujours suppléée par d'autres dispositions. Chez les Alphéidés, cette sup-. Fig. 113. Amphibetœus Jousseaimiei, R. Goutière, somites I, II, III (type). — Fig. 114. — Athanas nitescens^ Leach, antennule. — Fig. 115. Synalpheus mi- nor, Say, antennule. — Fig. 116. S. lœvimanus, var. longicarpus, Herrick, an- tennule (cotype). — Fig. 117. Alpheus armillatus, Milne-Edwar
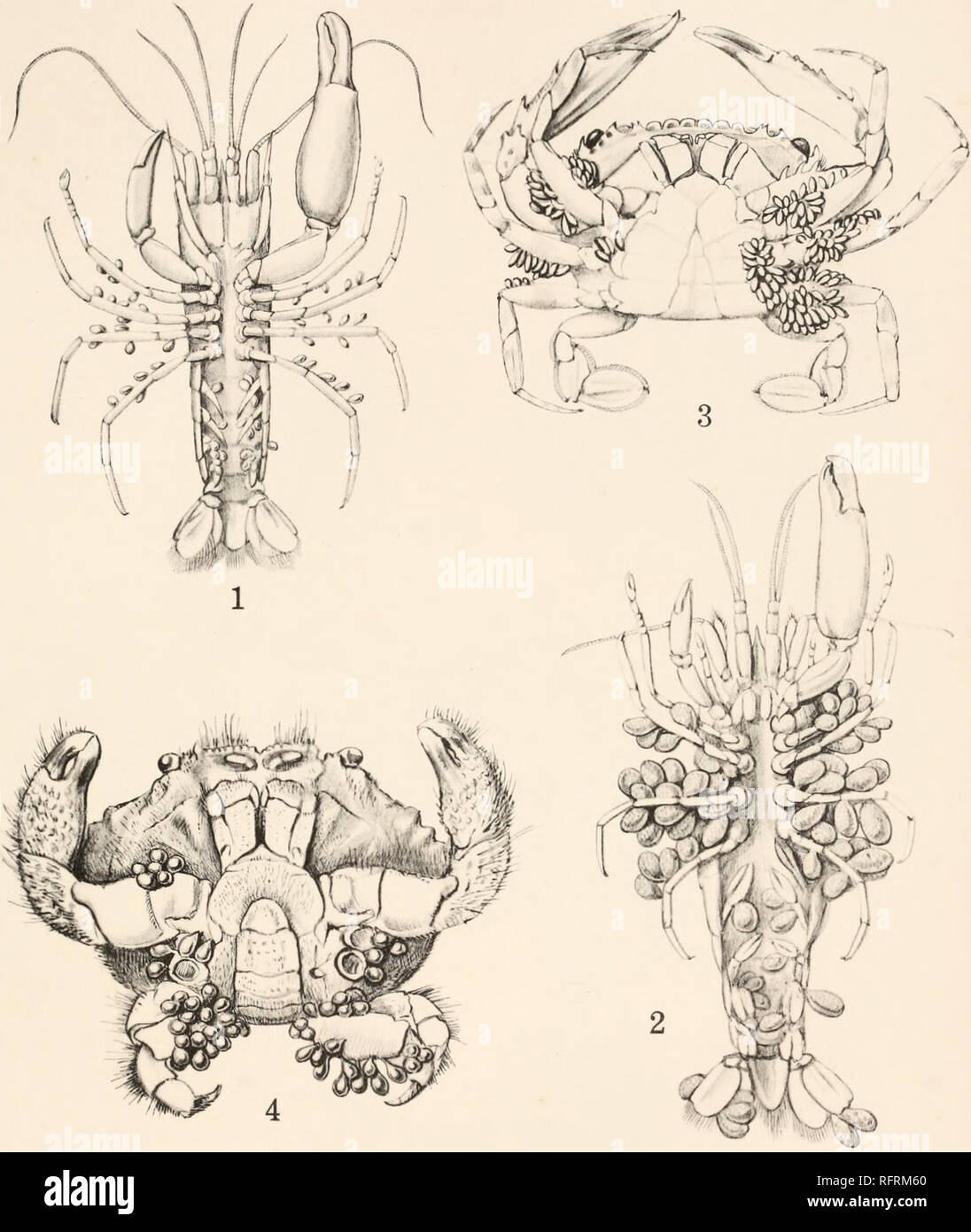
RMRFRM60–. Carnegie Institution of Washington publication. F. A. POTTS PLATE 1. Fig. 1. Thompsonia on Synalpheus brucei. Young external sacs on the abdominal and thoracic appendages, x 3. Fig. 2. The same at a later stage. The external sacs now contain Cypris larvae which are roughly indicated within, x 3. Fig. 3. Thompsonia on Thalamita prymna. To show the very large number of external sacs occurring on this specimen. Natural size. Fig. 4. Thompsonia on Actasa ruppellii. External sacs pear-shaped, x 2. The first three specimens figured are from Murray Island, the fourth from South Africa.. Please note
RMRMYA2F–. Annales des sciences naturelles. Zoology; Biology. ALPHEID^. 353 les affinités entre les divers genres qui précèdent est le suivant : Alplieus Racilius Synalpheus Betœus Parabetseus. Amphibetœus Jousseaumea Alpheopsis Athanas uitescens Automate Athanopsis Ath. dimorph. Cheiro- et Djiboutensis thrix Pterocariî Ogyris En ce qui concerne la place des Alphéidés sur l'arbre phylogénétique des Eucyphotes, Boas et Ortmann ont donné deux schémas un peu différents, que j'ai reproduits antérieurement l'un et l'autre (Cli. i, p. 33 et 45). Je me bornerai à faire remarquer que le nombre des formes exami
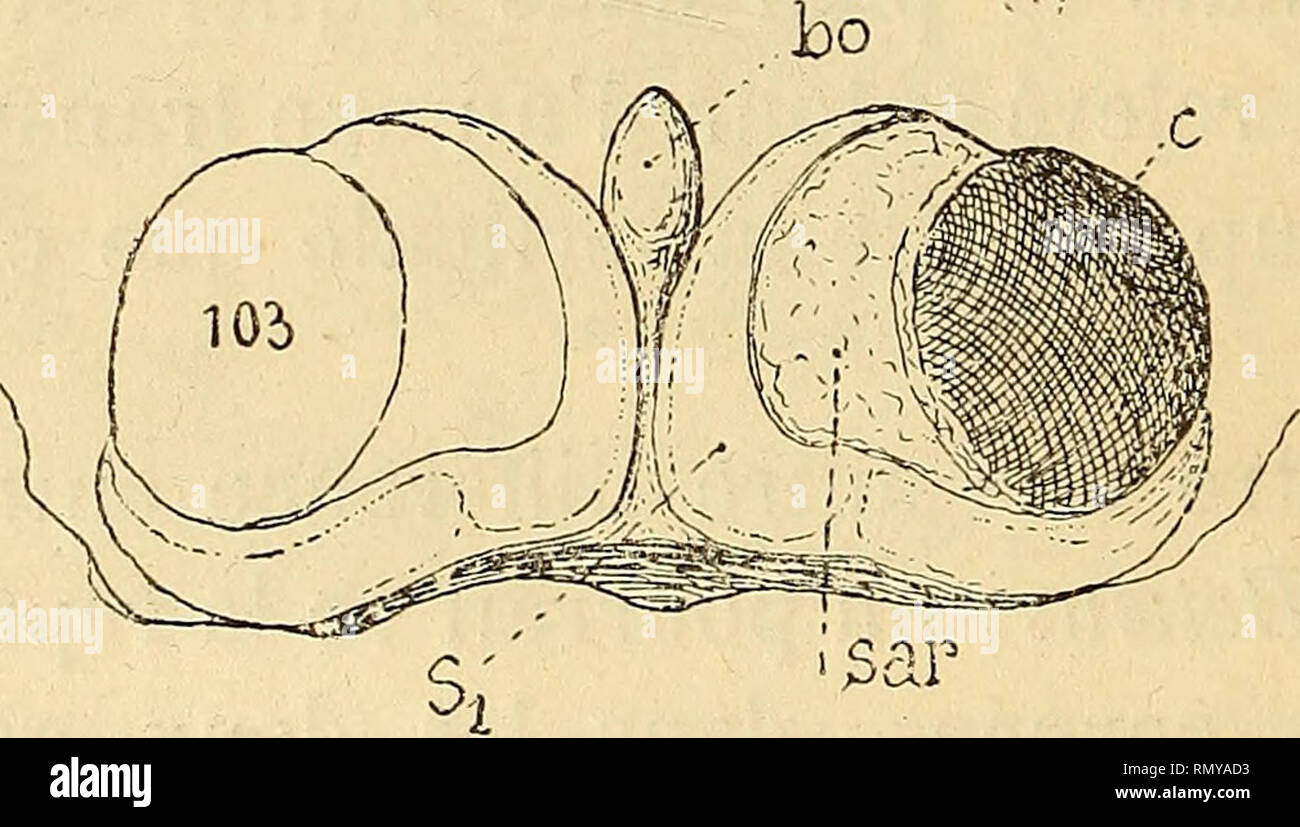
RMRMYAD3–. Annales des sciences naturelles. Zoology; Biology. 'Si :sâr. Fig. 98. Synalpheus mlnor, Say, soinite ophtalmique. — Fig. 99. .S. nepiunuSy Dana, somite oplitalmique. — Fig. 102. Alpheus pachychirus, Stimpson, somite ophtalmique. — Fig. 103. A. strejiuus, Dana, somite ophtalmique. — Fig. 104. A. megacheles, Hailstone, somite ophtalmique. vertical inférieur émis par la base du rostre (tig. 2^^prb) qui vient à la rencontre du bec ocellaire et s'échancre même pour recevoir celui-ci. La protection de la région médiane est ainsi assurée, aussi les saillies des ophtalmopodes sont- elles plus faible

RMRMYA9F–. Annales des sciences naturelles. Zoology; Biology. 166 H. COUTlEUi:. Boas a fait ressortir le caractère distinctif résultant, pour les Eucyphotes, d'un prolongement latéral de Texopodite, en forme de crête foliacée, qu'il désigne par la lettre a. Ce. Fig. ISl. Synalpheus minoi% Say, maxillipède I, lacinie interne. — Fig. 182. ^m- phibetœùs Jousseuumei, H. Coulière, lacinie interne. — Fig. 183. Hippolyte gib- berosiis, M.-Edwards, lacinie interne. — Fig. 184. Alpheus deuteropus, Hilgen- dorf, maxillipède I, endopodite ou « palpe ». — Fig. 184 bis. Alpheus malleu- tor, Dana, id. — Fig. 185. A.
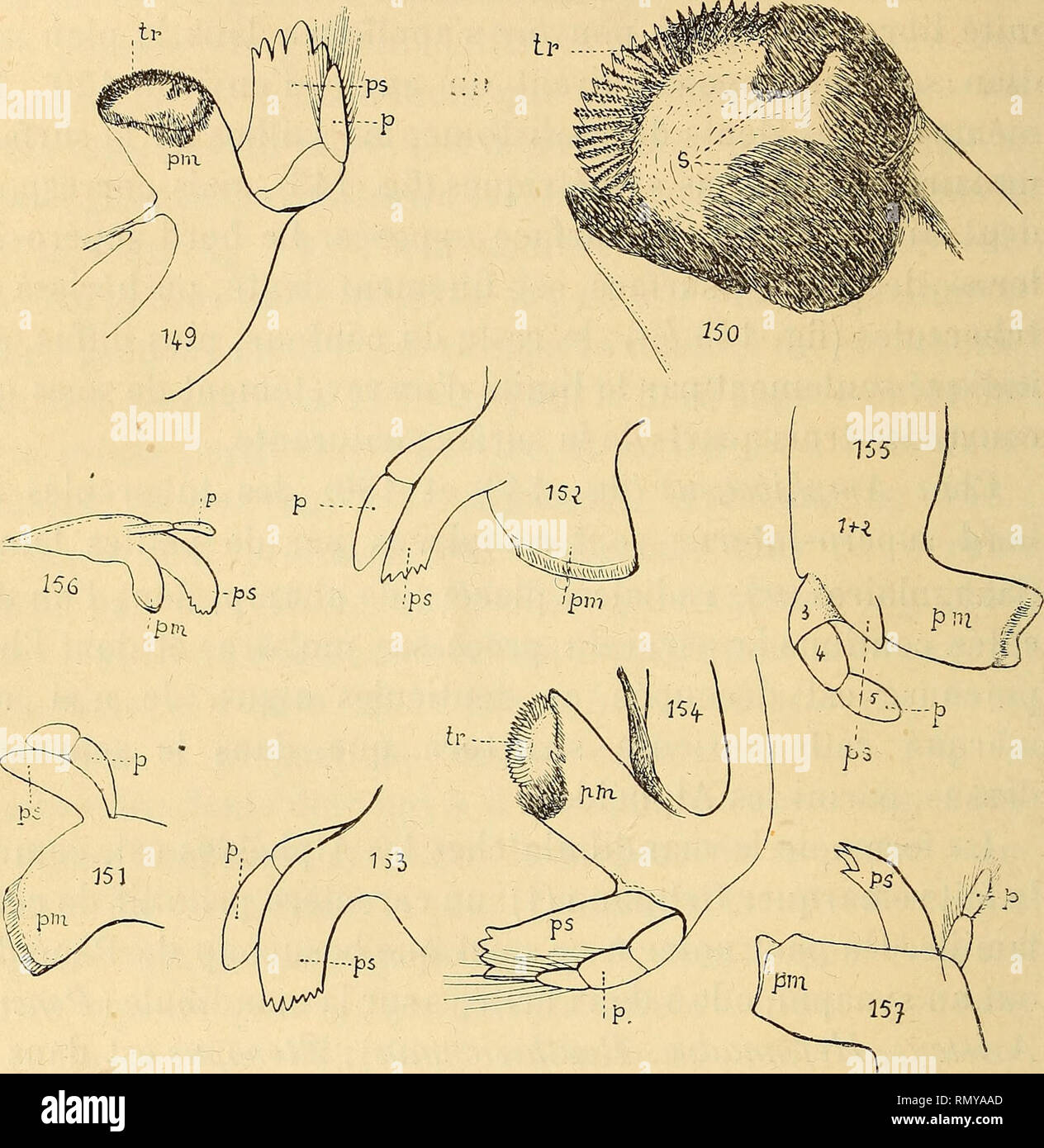
RMRMYAAD–. Annales des sciences naturelles. Zoology; Biology. 156 H. COUTIEUE. phipode a le plus souvent trois articles chez les Hippolyli- dés (fîg. 154), comme aussi chez les Palémonidés, lorsqu'il existe ; mais il en a deux seulement chez Hipp. polaris^ et de. Fig. 149. Amphibetœus Jousseautnei, H. Coutière, mandibule (type). — Fig. 150. Jd., détails du processus molaire. — Fig. 151. Synalpheus CGmatularum, Hass- well, mandibule (cotype). — Fig. 152. S. Stimpsoni, de Man, mandibule. — Fig. 153. S, laevimanus, var. longicarpus, Herrick, mandibule (type). — Fig. 154. — Hipp. gibberosus, M.-Edwards, ma
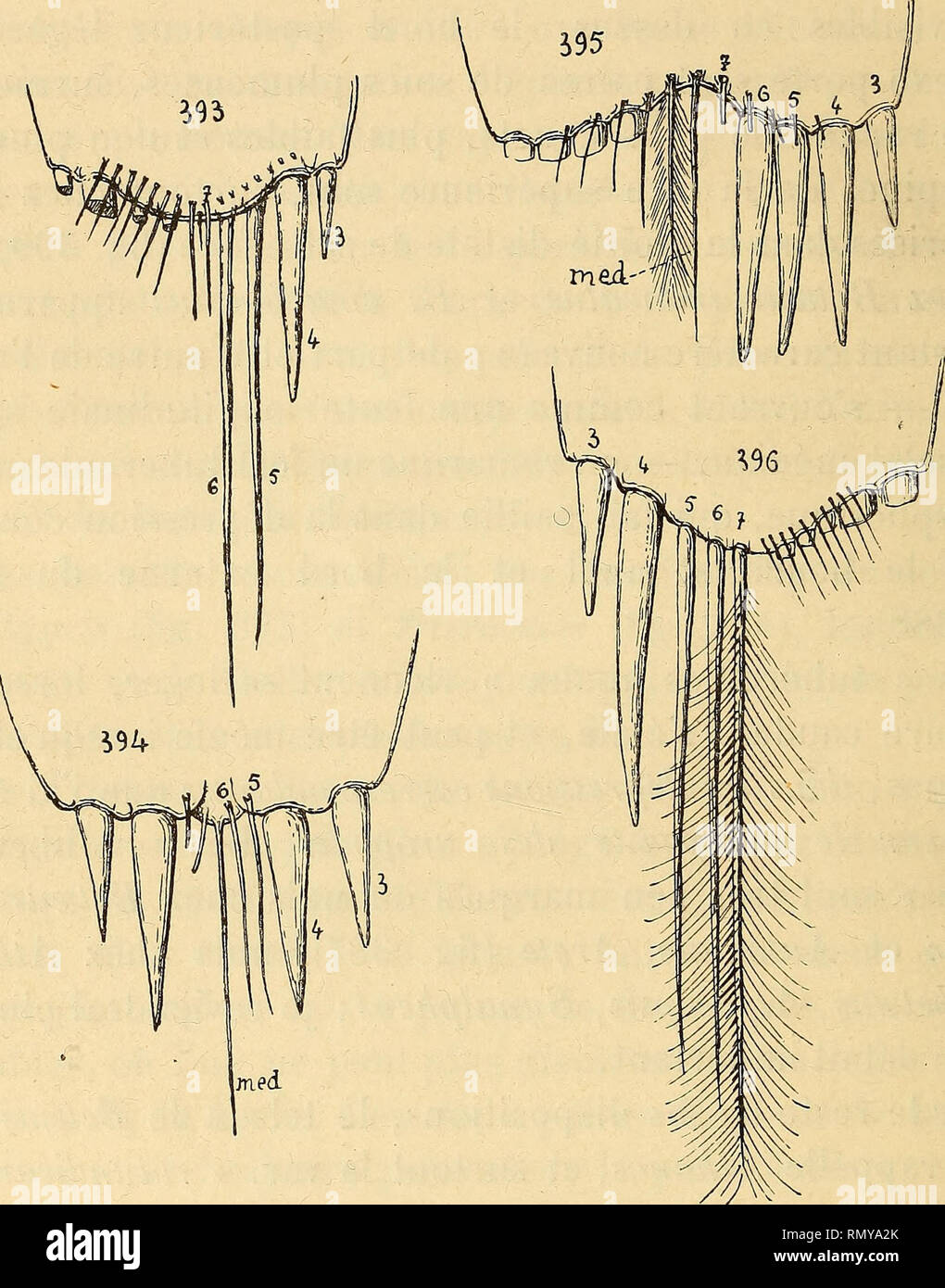
RMRMYA2K–. Annales des sciences naturelles. Zoology; Biology. 314 IT. COUTIERE. enfin chacune de ses moitiés porte quatre fortes épines au lieu de deux, et deux paires de soies plumeuses plus internes. L'une de celles-ci appartient sans doute à la rangée des poils, au nombre de 4-5, qui surmontent l'insertion des. Fig. 393. Automate doUchognatha^ de Man^ telson, armature distale. — Fig. 39L Synalpheus lœvimanus, var. longica7'pus, Rerricyi (id.).— Fig. 395. Jou^seaumea serratidigitiis, H. Coutière (id.) (type). — |^ig. 396. Alpheopsis trispinosus, Stimpson (id.). épines. 11 y a, comme à l'ordinaire, de
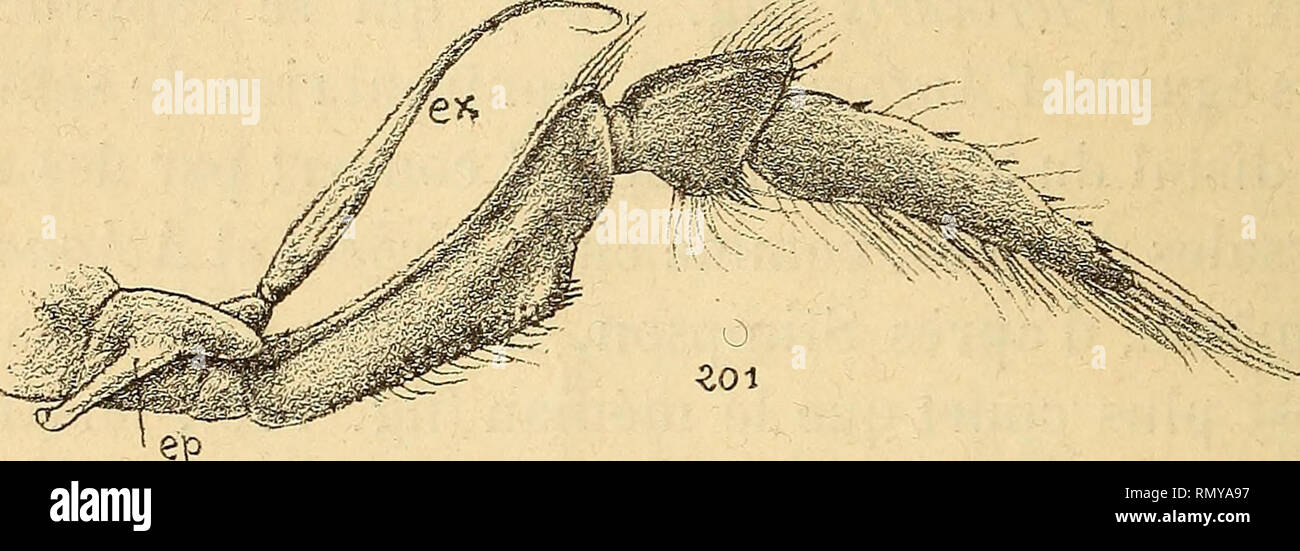
RMRMYA97–. Annales des sciences naturelles. Zoology; Biology. 172 II. COUTiKRE. forme liabitLielle des maxillipèdes lïl chez les Alpliéidés. 1 Synalpheus comatularum^ S. Stïmpsom^ S. carinatus sont dans ce dernier cas. Il est bon de remarquer que ces espèces de Synalpheus sont, par plusieurs points, celles qui sont le moins « alphéi- formes », comme j'aurai occasion de le faire ressortir par ailleurs. De même, Ogyris et Pterocarïs sont moins évolués dans ce sens qu'Automate; enfin Atha?ias est dans le même cas vis-à-vis à'Alpheus. Il se trouve donc que les formes alphéennes les plus primitives ont l'ar
Search Results for Synalpheus Stock Photos and Images (105)
Page 1 of 2