Post by Life on Mar 21, 2021 0:56:40 GMT 5
Sperm Whale (Physeter macrocephalus)

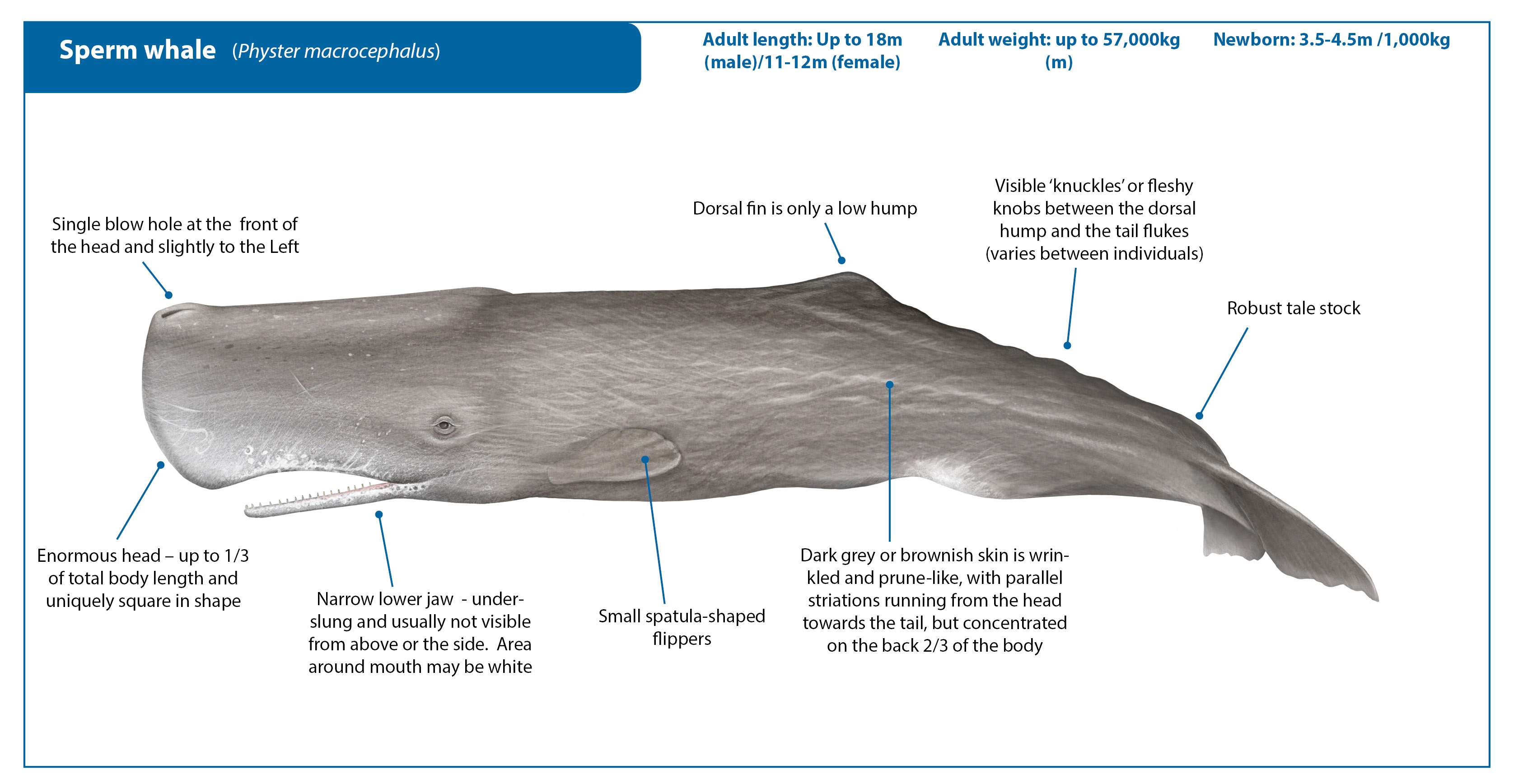

Documented sizes
From McClain et al (2015) for reference:
Citation: McClain, C. R., Balk, M. A., Benfield, M. C., Branch, T. A., Chen, C., Cosgrove, J., ... & Thaler, A. D. (2015). Sizing ocean giants: patterns of intraspecific size variation in marine megafauna. PeerJ, 3, e715. LINK: peerj.com/articles/715/?utm_content=buffer3b7cb&utm_medium=social&utm_source=twitter.com&utm_campaign=buffer


Citation: Miller, P. J., Aoki, K., Rendell, L. E., & Amano, M. (2008). Stereotypical resting behavior of the sperm whale. Current biology, 18(1), R21-R23. LINK: www.sciencedirect.com/science/article/pii/S0960982207022002

Picture courtesy Gabriel Barathieu
Classification
| Kingdom | Animalia |
| Phylum | Chordata |
| Subphylum | Vertebrata |
| Class | Mammalia |
| Order | Cetacea |
| Family | Phyeteridae |
| Genus | Physeter |
| Species | Phyeter catodon |
| Niche | Carnivore |
| Habitat | Benthic (Dive duration: 90 - 140 minutes) |
---
General references
Animal Diversity: animaldiversity.org/accounts/Physeter_catodon/
National Geographic: www.nationalgeographic.com/animals/mammals/facts/sperm-whale
Oceanwide Expeditions: oceanwide-expeditions.com/to-do/wildlife/sperm-whale
---
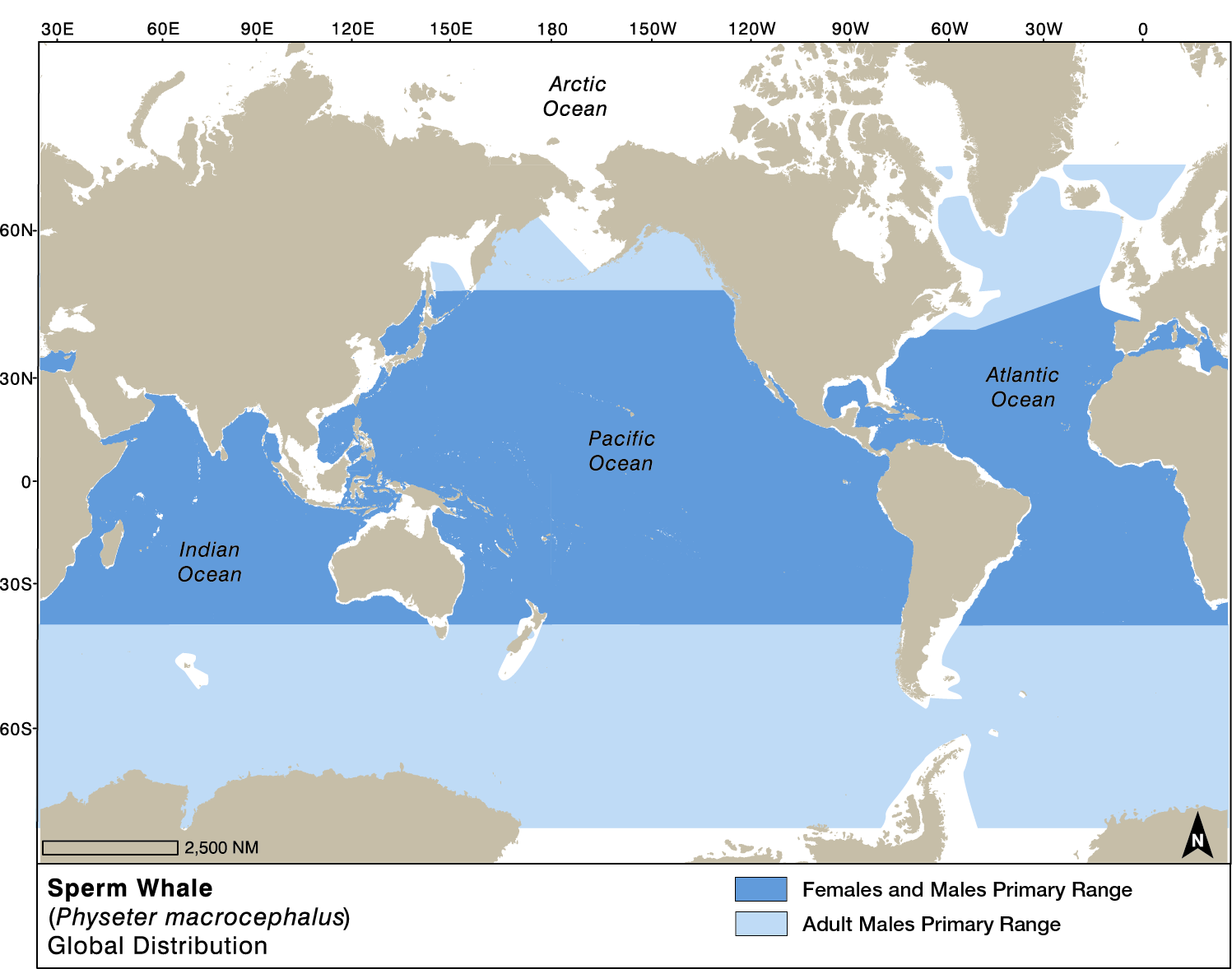
Global Range

LINK: iwc.int/sperm-whale
-----
Physiology and Anatomy

LINK: iwc.int/sperm-whale
---
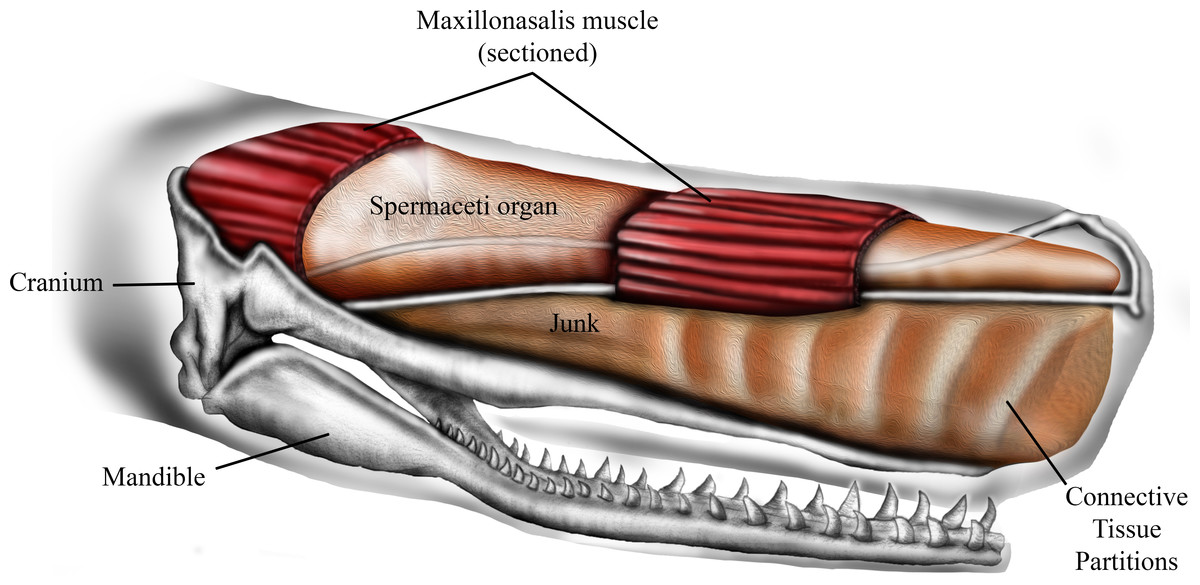
Forehead

Figure 1: Schematic representation of sperm whale head structure. Image courtesy of Ali Nabavizadeh.
Herman Melville’s novel Moby Dick was inspired by historical instances in which large sperm whales (Physeter macrocephalus L.) sank 19th century whaling ships by ramming them with their foreheads. The immense forehead of sperm whales is possibly the largest, and one of the strangest, anatomical structures in the animal kingdom. It contains two large oil-filled compartments, known as the “spermaceti organ” and “junk,” that constitute up to one-quarter of body mass and extend one-third of the total length of the whale. Recognized as playing an important role in echolocation, previous studies have also attributed the complex structural configuration of the spermaceti organ and junk to acoustic sexual selection, acoustic prey debilitation, buoyancy control, and aggressive ramming. Of these additional suggested functions, ramming remains the most controversial, and the potential mechanical roles of the structural components of the spermaceti organ and junk in ramming remain untested. Here we explore the aggressive ramming hypothesis using a novel combination of structural engineering principles and probabilistic simulation to determine if the unique structure of the junk significantly reduces stress in the skull during quasi-static impact. Our analyses indicate that the connective tissue partitions in the junk reduce von Mises stresses across the skull and that the load-redistribution functionality of the former is insensitive to moderate variation in tissue material parameters, the thickness of the partitions, and variations in the location and angle of the applied load. Absence of the connective tissue partitions increases skull stresses, particularly in the rostral aspect of the upper jaw, further hinting of the important role the architecture of the junk may play in ramming events. Our study also found that impact loads on the spermaceti organ generate lower skull stresses than an impact on the junk. Nevertheless, whilst an impact on the spermaceti organ would reduce skull stresses, it would also cause high compressive stresses on the anterior aspect of the organ and the connective tissue case, possibly making these structures more prone to failure. This outcome, coupled with the facts that the spermaceti organ houses sensitive and essential sonar producing structures and the rostral portion of junk, rather than the spermaceti organ, is frequently a site of significant scarring in mature males suggest that whales avoid impact with the spermaceti organ. Although the unique structure of the junk certainly serves multiple functions, our results are consistent with the hypothesis that the structure also evolved to function as a massive battering ram during male-male competition.
+
Our results are not directly relevant to the behavioral strategies behind ramming impacts; however, our findings are consistent with the hypothesis proposed in 1821 by Owen Chase (Chase, 1821). Following the sinking of the Essex whaling ship, Owen Chase hypothesized that sperm whales not only use their immense and elaborately complex foreheads as battering rams when fighting, but also that “the whale’s head is admirably designed for this mode of attack.” The prevalence of head-butting in sperm whales is not well documented. However, ramming is a basal behavior for Bovidae (Farke, 2008; Alvarez, 1990) and Cetacea (Carrier, Deban & Otterstrom, 2002), including humpback whales (Baker & Herman, 1984), bottle-nosed whales (Gowans & Rendell, 1999), narwhales (Silverman & Dunbar, 1980), long-finned pilot whales (Reilly & Shane, 1986) and killer whales (Goley & Straley, 1994). Based on these reports, it has previously been hypothesized that the spermaceti organ of male sperm whales may function as a weapon and is more developed in males due to sexual selection (Carrier, Deban & Otterstrom, 2002). If this is true, then males may be exposed to increased stresses during head-butting ramming and as such necessitate additional support via a dramatically increased and more structurally robust melon.
Citation: Panagiotopoulou, O., Spyridis, P., Abraha, H. M., Carrier, D. R., & Pataky, T. C. (2016). Architecture of the sperm whale forehead facilitates ramming combat. PeerJ, 4, e1895. LINK: peerj.com/articles/1895/
The Essex whaling ship
---
Acoustics
Sperm whales can hunt in total darkness under the sea but how? Steve Backshall explains how sperm whales use sound to build up a picture of their environment as he gets up close to them.
From Fais et al (2016) for reference:
The sperm whale carries a hypertrophied nose that generates powerful clicks for long-range echolocation. However, it remains a conundrum how this bizarrely shaped apex predator catches its prey. Several hypotheses have been advanced to propose both active and passive means to acquire prey, including acoustic debilitation of prey with very powerful clicks. Here we test these hypotheses by using sound and movement recording tags in a fine-scale study of buzz sequences to relate the acoustic behaviour of sperm whales with changes in acceleration in their head region during prey capture attempts. We show that in the terminal buzz phase, sperm whales reduce inter-click intervals and estimated source levels by 1–2 orders of magnitude. As a result, received levels at the prey are more than an order of magnitude below levels required for debilitation, precluding acoustic stunning to facilitate prey capture. Rather, buzzing involves high-frequency, low amplitude clicks well suited to provide high-resolution biosonar updates during the last stages of capture. The high temporal resolution helps to guide motor patterns during occasionally prolonged chases in which prey are eventually subdued with the aid of fast jaw movements and/or buccal suction as indicated by acceleration transients (jerks) near the end of buzzes. - Fais et al (2016)
Figure 3

(A) Sperm whale shallow foraging dive profile with four prey capture attempts, i.e. buzzes, indicated by red circles. The black line indicates the echolocation phase of the dive. (B–D) represent different parameters from the start of the buzz to the peak in normalized jerk, taken as a proxy for prey interception. (B) A detailed view of the click rate during the final buzz (encircled in black in (A)). ICIs, from 10 s before the buzz to the end of the buzz, are colour coded by their relative AOL (dBpp re max pp). The red line marks the estimated two-way-travel time (TWT) from the sound source to the presumed prey capture location calculated from the forward speed of the whale. Note the log scale on the TWT axis. (C) Normalized jerk signature during the same period and (D) distance between the front of the whale and the presumed prey interception location, derived from the forward swimming speed of the whale, calculated using the orientation-corrected depth rate method.
Here we have addressed several foraging hypotheses for sperm whales using high resolution on-animal measurements to uncover the movement and echolocation behaviour before and during prey capture. Sperm whales produce the most powerful biological sounds in the animal kingdom in order to echolocate prey at long ranges, but reduce acoustic outputs by several orders of magnitude when they are about their own body length from their prey. This behaviour is inconsistent with the big bang hypothesis indicating that the extreme nasal complex of sperm whales did not evolve to debilitate prey with sound, but rather to produce powerful clicks for long range echolocation of prey. There is likewise no indication that visual predation plays a significant role in sperm whale foraging. Instead, prey capture attempts by sperm whales are remarkably similar to those of other echolocating toothed whales comprising a sudden change in click rate and output level when the whales are about a body length from their prey. High repetition rate buzzes at low output levels provide high temporal and spatial resolution tracking to inform motor patterns in the last few meters before capture. While some prey targeted by sperm whales appear to be easy to subdue, others, often at shallower depths, require lengthy chases, leading to prolonged buzzes and higher than average swimming speeds. Strong and sudden changes in acceleration near the end of buzzes are supportive of the notion that sperm whales employ suction feeding to engulf their prey57 and continue buzzing during post-acquisition prey handling. Nonetheless, it still remains a conundrum how the sperm whale can keep track of a prey item during the final meters from when the prey drops below the sonar beam and until it reaches the buccal cavity, and we still do not understand the details of how the prey is engulfed. Hopefully, advances in animal-attached cameras will help answer these questions in the future. - Fais et al (2016)
Citation: Fais, A., Johnson, M., Wilson, M., Soto, N. A., & Madsen, P. T. (2016). Sperm whale predator-prey interactions involve chasing and buzzing, but no acoustic stunning. Scientific Reports, 6(1), 1-13. LINK: www.nature.com/articles/srep28562
---
Documented sizes
From McClain et al (2015) for reference:
Largest Odontocete: Sperm Whale, Physeter macrocephalus (Linnaeus, 1758)
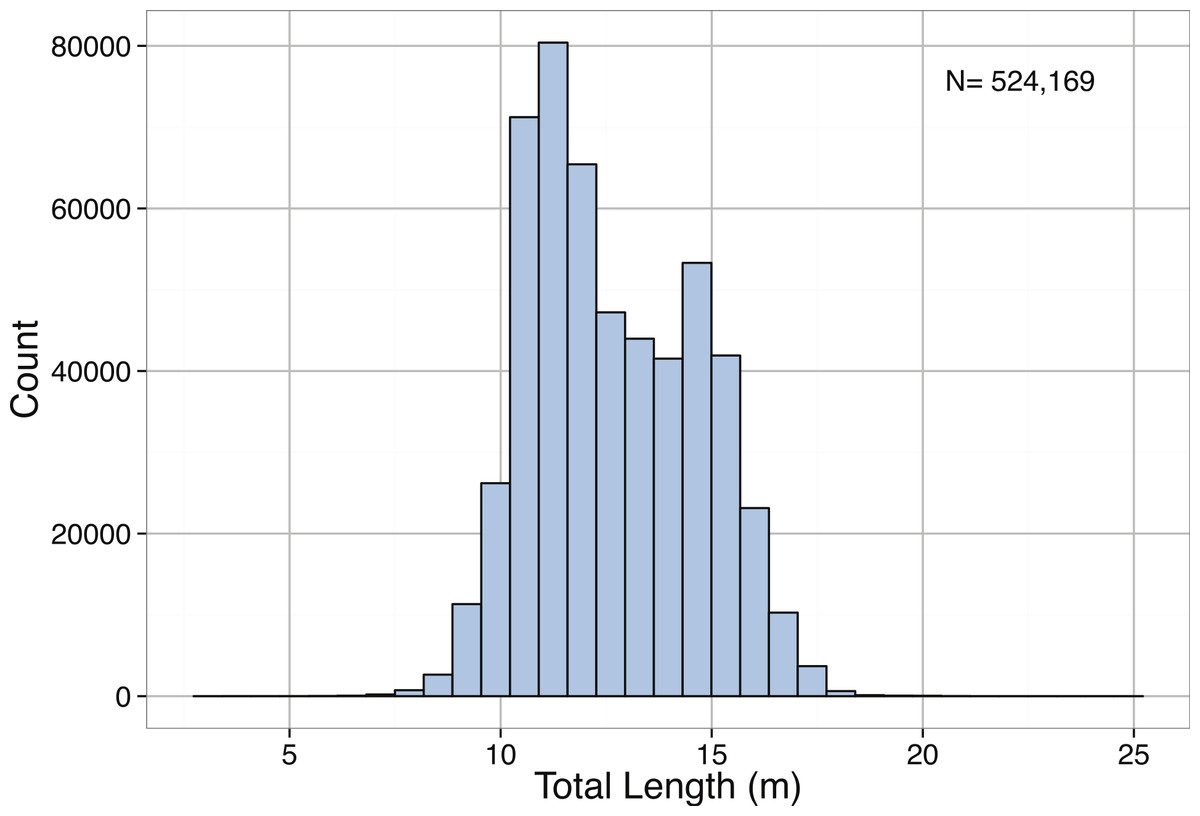
Physeter macrocephalus is the largest of the toothed whales. Sperm whales possess a cosmopolitan geographic distribution but, given their deep diving foraging behavior, tend to occur at depths greater than 1,000 m. The largest known individual, as reported by Guinness Records, was captured off the Kuril Islands in 1950 and measured 20.7 m in length (Carwardine, 1995). However, the jaw of the specimen in the Natural History Museum in London is purported to belong to a 25.6 m long individual (Carwardine, 1995). In our data set the longest measured length is 24 m, given for a male caught in the South Pacific in 1933. Even the next eight largest individuals (22.9–20.8 m) in our dataset are greater than the maximum length reported by Carwardine (1995) and recognized by Guinness Records.
Physeter macrocephalus is the largest of the toothed whales. Sperm whales possess a cosmopolitan geographic distribution but, given their deep diving foraging behavior, tend to occur at depths greater than 1,000 m. The largest known individual, as reported by Guinness Records, was captured off the Kuril Islands in 1950 and measured 20.7 m in length (Carwardine, 1995). However, the jaw of the specimen in the Natural History Museum in London is purported to belong to a 25.6 m long individual (Carwardine, 1995). In our data set the longest measured length is 24 m, given for a male caught in the South Pacific in 1933. Even the next eight largest individuals (22.9–20.8 m) in our dataset are greater than the maximum length reported by Carwardine (1995) and recognized by Guinness Records.
The large size of P. macrocephalus may be attributed to their foraging behavior (Evans & Hindell, 2004; Rice, 1989; Watwood et al., 2006; Whitehead, MacLeod & Rodhouse, 2003). As a known deep-sea diver, P. macrocephalus regularly dives to depths that few other pelagic animals reach (Watwood et al., 2006). Large size confers a benefit in these deep-sea excursions, not only by making the trip less metabolically costly, but also by increasing aerobic capacity and enabling P. macrocephalus to stay submerged for longer periods of time (Watwood et al., 2006; Whitehead, MacLeod & Rodhouse, 2003). Large size also allows P. macrocephalus to feed on many cephalopods during a single dive (Rice, 1989). Lindberg & Pyenson (2007) hypothesized that the wide diversity of cephalopods during the Eocene allowed archaeocetes, primitive cetaceans, to exploit mid- and deep-water cephalopods and subsequently evolve into large odontocetes capable of diving to great depths. However, cephalopods may provide a lower quality food source than fish and crustaceans (Evans & Hindell, 2004) and ultimately may energetically limit the maximum size obtainable by odontocetes.
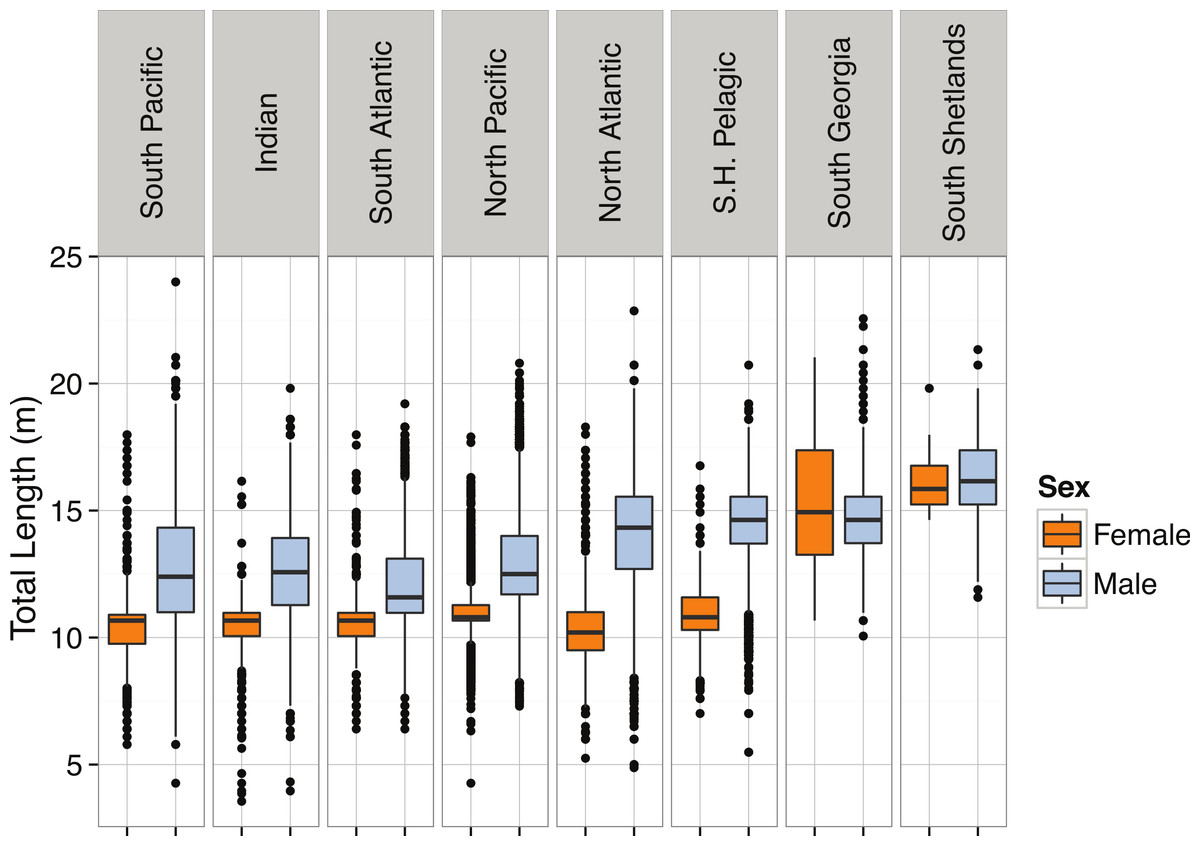
Size data obtained for P. macrocephalus came from the International Whaling Commission’s (IWC) whaling records (data held by IWC). Extending as far back as the 1880s, the records detail the ocean basin where individuals were caught, the specific geographical coordinates of capture, the date of capture, and the sex of each individual. The total lengths of individuals were measured from whales at rest on a flat surface, from the apex of the notch between tail flukes to the most forward part of the head. We found that the overall distribution of total lengths for P. macrocephalus was right-skewed (Table 3; Fig. 34). Geographic differences in the length of P. macrocephalus were found, with individuals in the seas of the Southern Ocean and North Atlantic obtaining larger mean lengths relative to individuals in other regions (Fig. 35). In the North Atlantic and Southern Hemisphere pelagic (open-ocean) whaling, the differences in sizes only occur in males, while at South Georgia and South Shetlands both males and females are larger. The wide variation in the geographic size distribution seen in P. macrocephalus is consistent with observations of the whale’s mating patterns and distribution. Individuals found closest to the poles are predominantly solitary large, mature males, while larger groups of females and immature males congregate closer to the equator (Best, 1979).
We also found total length to differ between sexes, with males being larger than females (p < 2 × 10−16, Fig. 35). Physeter macrocephalus are the most sexually dimorphic of the cetaceans, which has been attributed to sexual competition between males for mating opportunities (Rice, 1989; Whitehead, MacLeod & Rodhouse, 2003). Females stop growing at approximately 30 years of age and 10.6 m in length, while males continue growing until they are 50 years old and 16 m long. The sexes also demonstrate differences in geographic distributions, as mature males can be found in waters cooler than 15 °C at the surface, while females and immature males remain in tropical and sub-tropical regions (Best, 1979). Mature males only return to the warmer waters in order to breed (Best, 1979). The mean lengths of males and females were significantly different within all the geographic regions (from p = 0.0018 to p < 2 × 10−16), but relative differences varied between regions. Sexual dimorphism was the weakest among males and females of P. macrocephalus caught off South Georgia and South Shetlands, where many mature males and a few large females occur.
In 1999, it was estimated that the population of P. macrocephalus was only 32% of the pre-whaling population of 1,100,000 whales (Whitehead, 2002). However, Whitehead’s (2002) estimate of current sperm whale abundance may be too low, since it was assumed that 87% of sperm whales on the survey tracklines were observed (much higher than is realistic, T Branch, pers. comm., 2014). On the other hand, the current population size may be more depleted due to the revelation that the Soviet Union caught many more whales in the 1950s–1970s than they had previously reported (Baker & Clapham, 2004). The disproportionate number of male individuals harvested by whalers (except the later Soviet catches) suggests that they selectively took larger-bodied specimens from which more oil could be produced, a process known as “gunner selection” (Ellis, 2011).
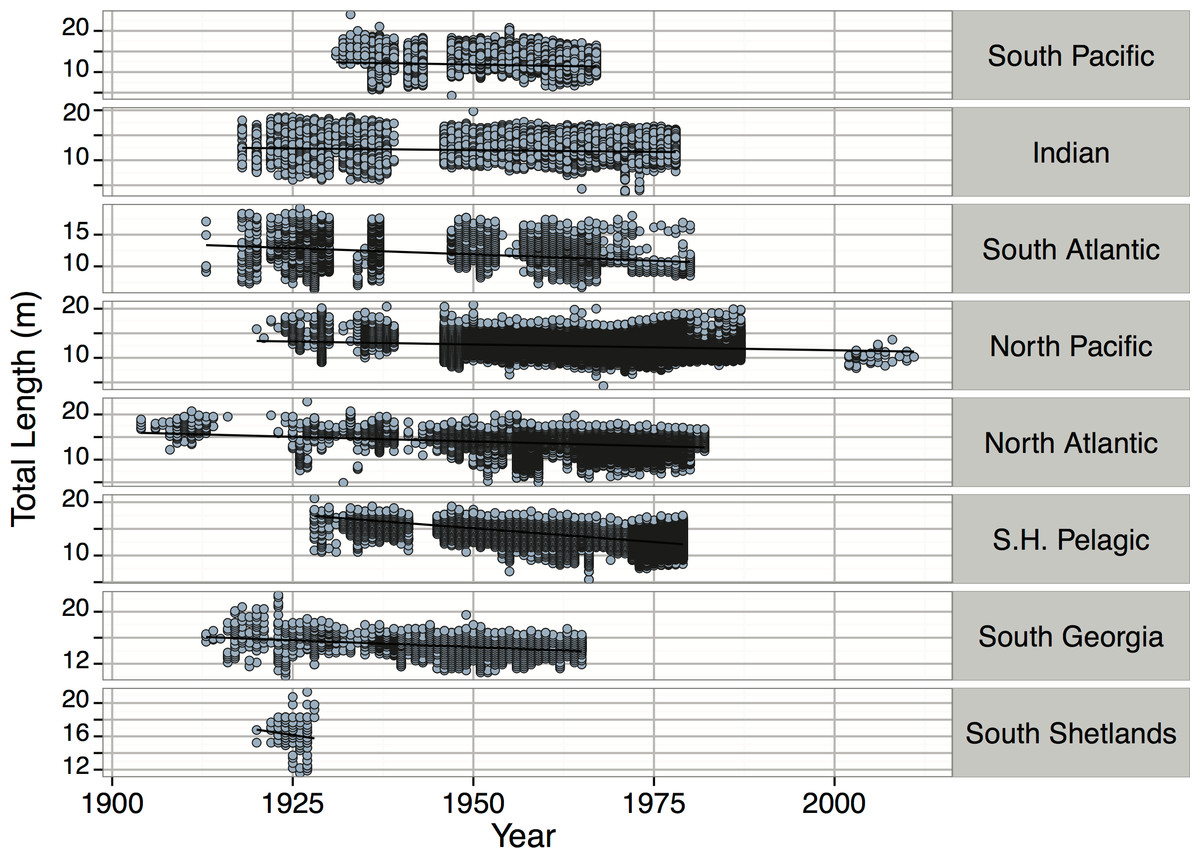
We found a significant temporal decline in the sizes of both males and females (p < 2 × 10−16, Fig. 36). This trend of decreasing body size over time was consistent for all regions (from p = 4.05 × 10−06 to p < 2 × 10−16), with the exception of the South Shetlands (p = 0.1932). Declines may be due to heavy whaling on larger individuals, but may also be due to a shift from targeting only large lone males to more indiscriminate targeting of immature males and females, especially during the later period of Soviet whaling (about 1958–1973) when misreporting was rife (Ivashchenko & Clapham, 2014). The lack of pattern in the South Shetlands may be due to relatively lower sample sizes. The only region with an increase in body size was the North Pacific, where the sizes of males caught before 1975 decreased as seen in the other regions, but actually increased from 1975 on. This pattern was probably driven by Soviet whaling in the North Pacific as described above. It has been estimated that the Soviet Union reported only slightly more than half the numbers of their actual catches to the IWC (Ivashchenko, Clapham & Brownell, 2011). Some of the larger whales that were caught were reported to be shorter in order to create a more convincing distribution between the largest whales and the smallest whales, which were reported as longer than they were in order to meet length minimums (Ivashchenko, Clapham & Brownell, 2011). Consequently, once observers were allowed onto whaling ships in 1972 (Tønnessen & Johnsen, 1982), the falsification of data lessened, resulting in the observed increase in whale size. - McClain et al (2015)
+
All of the species here tell a similar size tale, e.g., the maximum reported length for the sperm whale is 24 m but 95% of individuals measured are below 15 m and 75% are below 14.3 m. - McClain et al (2015)

Figure 34: Distribution of Total Length (m) for Physeter macrocephalus.

Figure 35: Boxplots of Total Length (m) by region and sex for Physeter macrocephalus.

Figure 36: Total Length (m) versus year by region for Physeter macrocephalus.
Check Table 3 in citation below.
Citation: McClain, C. R., Balk, M. A., Benfield, M. C., Branch, T. A., Chen, C., Cosgrove, J., ... & Thaler, A. D. (2015). Sizing ocean giants: patterns of intraspecific size variation in marine megafauna. PeerJ, 3, e715. LINK: peerj.com/articles/715/?utm_content=buffer3b7cb&utm_medium=social&utm_source=twitter.com&utm_campaign=buffer
-----
Napping behavior

A photographer recently snapped an image showing a group of whales sleeping vertically. (Image credit: Franco Banfi/Solent News & Photo Agency)
LINK: www.livescience.com/59910-sleeping-sperm-whales.html
Photographer Franco Banfi was free diving — underwater diving without a breathing apparatus — on Jan. 28 off the coast of Dominica, an island in the Caribbean Sea between Martinique and Guadalupe, when he spied six still and silent sperm whales drifting in their upright postures at a depth of around 65 feet (20 meters).
Researchers first saw this unusual sleep behavior in sperm whales in 2008, describing it in a study published in January of that year in the journal Current Biology. The scientists in that study found that sperm whales dozed in this upright drifting posture for about 10 to 15 minutes at a time, and the whales did not breathe or move at all during their naps, the study authors reported. [Sleep Tight! Snoozing Animals Gallery]
Researchers first saw this unusual sleep behavior in sperm whales in 2008, describing it in a study published in January of that year in the journal Current Biology. The scientists in that study found that sperm whales dozed in this upright drifting posture for about 10 to 15 minutes at a time, and the whales did not breathe or move at all during their naps, the study authors reported. [Sleep Tight! Snoozing Animals Gallery]
LINK: www.livescience.com/59910-sleeping-sperm-whales.html
From Miller et al (2008) for reference:
The vertical drifting posture recorded by the tags matches that of whales occasionally observed at sea (Figure 2). In an opportunistic experiment, video was recorded from R/V Balaena as it approached a group of 12–13 whales under sail alone (see Supplemental movie). Six whales can clearly be seen to be motionless in a vertical posture with the head at or just below the sea surface. Three vertical whales were unusually non-responsive to the very close (<2 m) approach of the vessel, reacting only when unintentionally touched by the vessel, after which the entire group moved away. In contrast, a horizontally logging whale in the group reacted to the vessel at ∼8 m distance. - Miller et al (2008)

Figure 2. Video frame showing three vertically drifting sperm whales passed within a few meters by R/V Balaena on 31 October 2000 off Northern Chile.
The vessel approached slowly with engines off from the left prior to this frame and continued moving to the right following this frame (blue arrow). The red arrows indicate the mid-line axes of the bodies of the drifting whales. Note that the whales on the left and right had visibility of the vessel with the right eye prior to this frame, and then with the left eye as the vessel passed. The whale in the centre was slightly off-axis so the right eye may not have had visibility of the vessel (see Supplemental movie for entire sequence).
The vessel approached slowly with engines off from the left prior to this frame and continued moving to the right following this frame (blue arrow). The red arrows indicate the mid-line axes of the bodies of the drifting whales. Note that the whales on the left and right had visibility of the vessel with the right eye prior to this frame, and then with the left eye as the vessel passed. The whale in the centre was slightly off-axis so the right eye may not have had visibility of the vessel (see Supplemental movie for entire sequence).
Check Supplemental movie in citation below.
Citation: Miller, P. J., Aoki, K., Rendell, L. E., & Amano, M. (2008). Stereotypical resting behavior of the sperm whale. Current biology, 18(1), R21-R23. LINK: www.sciencedirect.com/science/article/pii/S0960982207022002
-----
Up-close swimming and diving experiences
--- 2017 ---
--- 2015 ---
--- 2013 ---
--- 2012 ---