Abstract
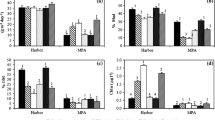
Clones of hermatypic coral Acropora digitifera from Sesoko Island, Okinawa, were exposed to temperatures of 29 °C, 30 °C, 31 °C, and 32 °C for 14 days in a laboratory to identify their response to high seawater temperature. Coral colour was evaluated by RGB (R, red; G, green; B, blue) values and a 6-point brightness/saturation scale coral colour reference card. The colour of A. digitifera shifted slightly toward the black end of the scale in the 29 °C and 30 °C treatments throughout the experiment. The maximum effective quantum yield (ΔF/Fm′) was stable at these temperatures treatments. By the end of the experiment at 31 °C, coral colour and ΔF/Fm′ were slightly altered in the direction of bleaching, but the coral was not bleached. This result indicates that A. digitifera can resist bleaching for at least 2 weeks at 31 °C which is 2 °C above the highest summertime seawater temperature in Okinawa. In the 32 °C treatment, while bleaching occurred approximately on day 3, the ΔF/Fm′ was reduced but increased again with fluctuations. The succession of ΔF/Fm′ after exposure to 32 °C differed from that in corals exposed to herbicides. Taken together, these results suggest that corals may keep the platform for primary production after bleaching under high temperature conditions.





Similar content being viewed by others
Change history
29 January 2023
The article is updated to remove the Data Availability Statement.
References
Ainsworth TD, Hoegh-Guldberg O, Heron SF, Skirving WJ, Leggat W (2008) Early cellular changes are indicators of pre-bleaching thermal stress in the coral host. J Exp Mar Biol Ecol 364:63–71
Baird AH, Bhagooli R, Ralph PJ, Takahashi S (2009) Coral bleaching: the role of the host. Trends Ecol Evol 24:16–20
Bell T, Nishida K, Ishikawa K, Suzuki A, Nakamura T, Sakai K, Ohno Y, Iguchi A, Yokoyama Y (2017) Temperature-controlled culture experiments with primary polyps of coral Acropora digitifera: Calcification rate variations and skeletal Sr/Ca, Mg/Ca, and Na/Ca ratios. Palaeogeogr Palaeoclimatol Palaeoecol 484:129–135
Brodie JE, Kroon FJ, Schaffelke B, Wolanski EC, Lewis SE, Devlin MJ, Bohnet IC, Bainbridge ZT, Waterhouse J, Davis AM (2012) Terrestrial pollutant runoff to the Great Barrier Reef: an update of issues, priorities and management responses. Mar Pollut Bull 65:81–100
Burke L, Reytar K, Spalding M, Perry A (2011) Reefs at Risk Revisited. World Resources Institute, Washington DC, pp i–x + 1–114
Gushi M, Ishibashi H, Takayama K, Yamashiro H, Takeuchi I (2021) Changes in the colour and photosynthetic efficiency of the hermatypic coral Acropora tenuis exposed to Irgarol 1051 at 30 °C seawater temperature. Reg Stud Mar Sci 47:101957
Hackerott S, Martell HA, Eirin-Lopez JM (2021) Coral environmental memory: causes, mechanisms, and consequences for future reefs. Trends Ecol Evol 36:1011–1023
Hayashi E, Suzuki A, Nakamura T, Iwase A, Ishimura T, Iguchi A, Sakai K, Okai T, Inoue M, Araoka D, Murayama S, Kawahata H (2013) Growth-rate influences on coral climate proxies tested by a multiple colony culture experiment. Earth Planet Sci Lett 362:198–206
Heron SF, Maynard JA, van Hooidonk R, Eakin CM (2016) Warming trends and bleaching stress of the world’s coral reefs 1985–2012. Sci Rep 6:38402. https://doi.org/10.1038/srep38402
Hirayama K, Takayama K, Haruta S, Ishibashi H, Takeuchi I (2017) Effect of low concentrations of Irgarol 1051 on RGB (R, red; G, green; B, blue) colour values of the hard-coral Acropora tenuis. Mar Pollut Bull 124:678–686
Hoeksema BW, Cairns S (2022) World list of Scleractinia. Acropora tenuis (Dana, 1846). World Register of Marine Species. http://www.marinespecies.org/aphia.php?p=taxdetails&id=207105. Accessed 04 Nov 2022
Hongo C, Yamano H (2013) Species-specific responses of corals to bleaching events on anthropogenically turbid reefs on Okinawa Island, Japan, over a 15-year period (1995–2009). PLoS ONE 8:e60952. https://doi.org/10.1371/journal.pone.0060952
Hughes TP, Kerry JT, Álvarez-Noriega M, Álvarez-Romero JG, Anderson KD, Baird AH, Babcock RC, Beger M, Bellwood DR, Berkelmans R, Bridge TC, Butler IR, Byrne M, Cantin NE, Comeau S, Connolly SR, Cumming GS, Dalton SJ, Diaz-Pulido G, Eakin CM, Figueira WF, Gilmour JP, Harrison HB, Heron SF, Hoey AS, Hobbs J-PA, Hoogenboom MO, Kennedy EV, Kuo C-Y, Lough JM, Lowe RJ, Liu G, McCulloch MT, Malcolm HA, McWilliam MJ, Pandolfi JM, Pears RJ, Pratchett MS, Schoepf V, Simpson T, Skirving WJ, Sommer B, Torda G, Wachenfeld DR, Willis BL, Wilson SK (2017) Global warming and recurrent mass bleaching of corals. Nature 543:373–377
Hughes TP, Kerry JT, Connolly SR, Baird AH, Eakin CM, Heron SF, Hoey AS, Hoogenboom MO, Jacobson M, Liu G, Skirving W, Pratchett MS, Sommer B, Torda G (2019) Ecological memory modifies the cumulative impact of recurrent climate extremes. Nat Clim Chang 9:40–43
Iguchi A, Suzuki A, Sakai K, Nojiri Y (2015) Comparison of the effects of thermal stress and CO2-driven acidified seawater on fertilization in coral Acropora digitifera. Zygote 23:631–634
Ishibashi H, Takaichi D, Takeuchi I (2021) Effects of the herbicide Irgarol 1051 on the transcriptome of hermatypic coral Acropora tenuis and its symbiotic dinoflagellates. Sci Total Environ 780:14652
Jokiel PL (2004) Temperature stress and coral bleaching. In: Rosenberg E, Loya Y (eds) Coral health and disease. Springer, Berlin, pp 401–425
Kamei M, Takayama K, Ishibashi H, Takeuchi I (2020) Effects of ecologically relevant concentrations of Irgarol 1051 in tropical to subtropical coastal seawater on hermatypic coral Acropora tenuis and its symbiotic dinoflagellates. Mar Pollut Bull 150:110734
Kayanne H (2017) Validation of degree heating weeks as a coral bleaching index in the northwestern Pacific. Coral Reefs 36:63–70
LaJeunesse TC, Parkinson JE, Gabrielson PW, Jeong HJ, Reimer JD, Voolstra CR, Santos SR (2018) Systematic revision of Symbiodiniaceae highlights the antiquity and diversity of coral endosymbionts. Current Biol 28:2570–2580
Liu G, Strong AE, Skirving W (2003) Remote sensing of sea surface temperatures during 2002 Barrier Reef coral bleaching. EOS Trans Am Geophys Union 84:137–144
Lough JM, van Oppen MJH (2009) Introduction coral bleaching—patterns, processes, causes and consequences. In: van Oppen MJH, Lough JM (eds) Ecological studies 203. Coral bleaching. Springer, Berlin, Heidelberg, pp 1–5
Loya Y, Sakai K, Yamazato K, Nakano Y, Sambali H, van Woesik R (2001) Coral bleaching: the winners and the losers. Ecol Lett 4:122–131
Manullang C, Millyaningrum IH, Iguchi A, Miyagi A, Tanaka Y, Nojiri Y, Sakai K (2020) Responses of branching reef corals Acropora digitifera and Montipora digitata to elevated temperature and pCO2. PeerJ 8:e10562. https://doi.org/10.7717/peerj.1056
Muko S, Suzuki G, Saito M, Nakamura T, Nadaoka K (2019) Transitions in coral communities over 17 years in the Sekisei Lagoon and adjacent reef areas in Okinawa, Japan. Ecol Res 34:524–534
Nakamura T (2017) Mass coral bleaching event in Sekisei lagoon observed in the summer of 2016. J Japan Coral Reef Soc 19:29–40
Nishiguchi S, Wada N, Yamashiro H, Ishibashi H, Takeuchi I (2018) Continuous recordings of the coral bleaching process on Sesoko Island, Okinawa, Japan, over about 50 days using an underwater camera equipped with a lens wiper. Mar Pollut Bull 131:422–427
Oakley CA, Davy SK (2018) Cell biology of coral bleaching. In: van Oppen MJH, Lough JM (eds) Ecological studies 233. Coral bleaching coral bleaching patterns, processes, causes and consequences. Springer International Publishing AG, Cham, pp 189–211
Ohki S, Kowalski RK, Kitanobo S, Morita M (2015) Changes in spawning time led to the speciation of the broadcast spawning corals Acropora digitifera and the cryptic species Acropora sp. 1 with similar gamete recognition systems. Coral Reefs 34:1189–1198
Quigley KM, Baker AC, Coffroth MA, Willis BL, van Oppen MJ (2018) Bleaching resistance and the role of algal endosymbionts. In: van Oppen MJ, Lough JM (eds) Ecological studies 233. Coral bleaching patterns, processes, causes and consequences. Springer International Publishing AG, Cham, pp 111–151
Sakai K, Singh T, Iguchi A (2019) Bleaching and post-bleaching mortality of Acropora corals on a heat-susceptible reef in 2016. PeerJ 7:e8138. https://doi.org/10.7717/peerj.8138
Shitaoka R, Ishibashi H, Takeuchi I (2021) Thermal tolerance of the hermatypic coral Acropora tenuis elucidated by RGB analysis and expression of heat shock proteins in coral and symbiotic dinoflagellates. Mar Pollut Bull 162:111812
Siebeck UE, Marshall NJ, Klüter A, Hoegh-Guldberg O, (2006) Monitoring coral bleaching using a colour reference card. Coral Reefs 25:453–460
Singh T, Iijima M, Yasumoto K, Sakai K (2019) Effects of moderate thermal anomalies on Acropora corals around Sesoko Island Okinawa. PLoS ONE 14:e0210795. https://doi.org/10.1371/journal.pone.0210795
Singh T, Sinniger F, Nakano Y, Nakamura S, Kadena S, Jinza M, Fujimura H, Harii S (2022) Long-term trends and seasonal variations in environmental conditions in Sesoko Island, Okinawa, Japan. Galaxea J Coral Reef Stud 24:121–133
Spalding MD, Brown BE (2015) Warm-water coral reefs and climate change. Science 350:769–771
Takeuchi I, Yamashiro H, Gushi M (2019) Usage of UV-curable oligomer-based adhesive agent in hermatypic coral experimental research. MethodsX 6:1600–1607
Takeuchi I, Takaichi D, Katsumata M, Ishibashi H (2020) Succession of delayed fluorescence correlated with coral bleaching in the hermatypic coral Acropora tenuis. Mar Pollut Bull 154:111008
van Dam JW, Negri AP, Uthicke S, Mueller JF (2011) Chemical pollution on coral reefs: exposure and ecological effects. In: Sánchez-Bayo F, van den Brink PJ, Mann RM (eds) Ecological impacts of toxic chemicals. Bentham Science Publishers, Sharjah, pp 87–211
Wooldridge SA (2009) Water quality and coral bleaching thresholds: Formalising the linkage for the inshore reefs of the Great Barrier Reef, Australia. Mar Pollut Bull 58:745–751
Yamano H, Satake K, Inoue T, Kadoya T, Hayashi S, Kinjo K, Nakajima D, Oguma H, Ishiguro S, Okagawa A, Suga S, Horie T, Nohara K, Fukuyama N, Hibiki A (2015) An integrated approach to tropical and subtropical island conservation. J Ecol Environ 38:271–279
Yorifuji M, Harii S, Nakamura R, Fudo M (2017) Shift of symbiont communities in Acropora tenuis juveniles under heat stress. PeerJ 5:e4055. https://doi.org/10.7717/peerj.4055
Acknowledgements
The sampling of Acropora was conducted under special permission from the Okinawa Prefectural Government (No. 31–13). The support of the Motobu Fisheries Cooperative Association is acknowledged for submitting the special permission. The present study was supported by a research grant from the Japan Society for the Promotion of Science (JSPS) Grants-in-Aid for Scientific Research (KAKENHI; grant nos. 18H03387). The authors express their thanks to two anonymous reviewers and the corresponding editor of Fisheries Science for useful comments. The authors would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Contributions
IT Conceptualization, Funding acquisition, Methodology, Project administration, Validation; Visualization, Writing–original draft, and Writing–review/editing. MM Data curation, Formal analysis, Investigation, Methodology, and Writing–original draft. HI Data curation, Formal analysis, Methodology, and Writing–review/editing. KT Methodology, Formal analysis. HY Investigation, Methodology.
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Takeuchi, I., Mizuguchi, M., Ishibashi, H. et al. Upper thermal tolerance of hermatypic coral Acropora digitifera collected from Sesoko Island, southern Japan, based on a laboratory experiment. Fish Sci 89, 181–189 (2023). https://doi.org/10.1007/s12562-022-01657-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12562-022-01657-3