Abstract
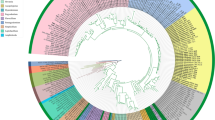
The subclass Chaetothyriomycetidae (Eurotiomycetes, Ascomycota) is an assemblage of ecologically diverse species, ranging from mutualistic lichenised fungi to human opportunistic pathogens. Recent contributions from molecular studies have changed our understanding of the composition of this subclass. Among others, ant-associated fungi, deep-sea fungi and bryophilous fungi were also shown to belong to this group of ascomycetes. The delimitation of orders and families within this subclass has not previously been re-assessed using a broad phylogenetic study and the phylogenetic position of some taxa such as the lichenised family Celotheliaceae or the Chaetothyrialean bryophilous fungi is still unclear. In our study, we assemble new and published sequences from 132 taxa and reconstruct phylogenetic relationships using four markers (nuLSU, nuSSU, mtSSU and RPB1). Results highlight several shortfalls in the current classification of this subclass, mainly due to un-assigned paraphyletic taxa. The family Epibryaceae is therefore described to circumscribe a previously un-assigned lineage. Celotheliales ad int. is suggested for the lineage including the lichen genus Celothelium and various plant pathogens. The delimitation of the family Trichomeriaceae is also broadened to include the genus Knufia and some anamorphic taxa. As defined here, Chaetothyriomycetidae includes four orders (Celotheliales ad int., Chaetothyriales, Pyrenulales, and Verrucariales) and ten families (Adelococcaceae, Celotheliaceae, Chaetothyriaceae, Cyphellophoraceae, Epibryaceae fam. nov., Herpotrichiellaceae, Pyrenulaceae, Requienellaceae, Trichomeriaceae, and Verrucariaceae).


Similar content being viewed by others
References
Aguirre-Hudson B (1991) A taxonomic study of the species referred to the ascomycete genus Leptorhaphis. Bull Br Mus Nat Hist Bot 21:85–192
Aptroot A (1991) A monograph of the Pyrenulaceae (excluding Anthracothecium and Pyrenula) and the Requienellaceae, with notes on the Pleomassariaceae, the Trypetheliaceae and Mycomicrothelia (lichenised and non-lichenised ascomycetes). Bibl Lichenologist 44:1–178
Aptroot A, Lücking R, Sipman HJM, Umaña L, Chaves JL (2008) Pyrenocarpous lichens with bitunicate asci: a first assessment of the lichen biodiversity inventory in Costa Rica. Bibliotheca Lichenologica No. 97. J. Cramer in der Gebrüder Borntraeger Verlagsbuchhandlung, Berlin and Stuttgart
Badali H, Gueidan C, Najafzadeh MJ, Bonifaz A, Gerrits van den Ende AHG, de Hoog GS (2008) Biodiversity of the genus Cladophialophora. Stud Mycol 61:175–191
Badali H, Prenafeta-Boldu FX, Guarro J, Klaassen CH, Meis JF, de Hoog GS (2011) Cladophialophora psammophila, a novel species of Chaetothyriales with a potential use in the bioremediation of volatile aromatic hydrocarbons. Fungal Biol 115:1019–1029
Batista AC, Ciferri R (1962) The Chaetothyriales. Sydowia 3:1–129
Berbee ML (1996) Loculoascomycete origins and evolution of filamentous ascomycete morphology based on 18S rRNA gene sequence data. Mol Biol Evol 13:462–470
Booth C, Ting WP (1964) Dolabra nepheliae gen. nov., sp. nov., associated with canker of Nephelium lappaceum. Trans Br Mycol Soc 47:235–237
Burgaud G, Le Calvez T, Arzur D, Vandenkoornhuyse P, Barbier G (2009) Diversity of culturable marine filamentous fungi from deep-sea hydrothermal vents. Environ Microbiol 11:1588–1600
Chomnunti P, Schoch CL, Aguirre-Hudson B, Ko Ko TW, Hongsanan S, Jones EBG, Kodsueb R, Phookamsak R, Chukeatirote E, Bahkali AH, Hyde KD (2011) Capnodiaceae. Fungal Divers 51:103–134
Chomnunti P, Bhat DJ, Jones EBG, Chukeatirote E, Bahkali AH, Hyde KD (2012a) Trichomeriaceae, a new sooty mould family of Chaetothyriales. Fungal Divers 56:63–76
Chomnunti P, Ko Ko TW, Chukeatirote E, Hyde KD, Cai L, Jones EBG, Kodsueb R, Hassan BA, Chen H (2012b) Phylogeny of Chaetothyriaceae in northern Thailand including three new species. Mycologia 104:382–395
Crous PW, Schubert K, Braun U, de Hoog GS, Hocking AD, Shin HD, Groenewald JZ (2007) Opportunistic, human-pathogenic species in the Herpotrichiellaceae are phenotypically similar to saprobic or phytopathogenic species in the Venturiaceae. Stud Mycol 58:185–217
Damm U, Fourie PH, Crous PW (2010) Coniochaeta (Lecythophora), Collophora gen. nov. and Phaeomoniella species associated with wood necroses of Prunus trees. Persoonia 24:60–80
Davey ML, Currah RS (2006) Interactions between mosses (Bryophyta) and fungi. Can J Bot 84:1509–1519
De Hoog GS, Guarro J, Gené J, Figueras MJ (2000) Atlas of Clinical Fungi, 2nd ed. Centraalbureau voor Schimmelcultures, Utrecht, Universitat Rovira i Virgili, Reus
Del Prado R, Schmitt I, Kautz S, Palice Z, Lücking R, Lumbsch HT (2006) Molecular data place Trypetheliaceae in Dothideomycetes. Mycol Res 110:511–520
Defossez E, Dubois MP, Mondolot L, Faccio A, Djieto-Lordon C, Mckey D (2009) Ant-plants and fungi: a new threeway symbiosis. New Phytol 1978:942–949
Diederich P, Ertz D, Lawrey JD, Sikaroodi M, Untereiner WA (2013) Molecular data place the hyphomycetous lichenicolous genus Sclerococcum close to Dactylospora (Eurotiomycetes) and S. parmeliae in Cladophialophora (Chaetothyriales). Fungal Divers 58:61–72
Döbbeler P (1978) Moosbewohnende Ascomyceten I. Die pyrenocarpen, den Gametophyten besiedelnden Arten. Mitt Bot Staatssamml München 14:1–360
Döbbeler P (1997) Biodiversity of bryophilous ascomycetes. Biodivers Conserv 6:721–738
Döbbeler P (2002) Microniches occupied by bryophilous ascomycetes. Nova Hedwigia 75:275–306
Dowell A (2001) Prosyllabus Tracheophytorum. Geos, Moscow
Drummond AJ, Rambaut A (2007) BEAST: bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7:214
Eriksson OE (1999) Outline of Ascomycota - 1999. Myconet 3:1–88
Eriksson OE (2006) Outline of Ascomycota - 2006. Myconet 12:1–82
Eriksson OE, Baral H-O, Currah RS, Hansen K, Kurtzman CP, Rambold G, Laessøe T (2004) Outline of Ascomycota - 2004. Myconet 10:1–99
Felix H (1988) Fungi on bryophytes, a review. Bot Helv 98:239–269
Feng B, Wang X, Hauser M, Kauffmann S, Jentsch S, Haase G, Becker JM, Szaniszlo PJ (2001) Molecular cloning and characterization of WdPKS1, a gene involved in dihydroxynaphthalene (DHN) melanin biosynthesis and virulence in Wangiella (Exophiala) dermatitidis. Infect Immun 69:1781–1794
Gardes M, Bruns TD (1993) ITS primers with enhanced specificity for Basidiomycetes: application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Geiser DM, Gueidan C, Miadlikowska J, Lutzoni F, Kauff F, Hofstetter V, Fraker E, Schoch CL, Tibell L, Untereiner WA, Aptroot A (2006) Eurotiomycetes: Eurotiomycetidae and Chaetothyriomycetidae. Mycologia 98:1053–1064
Gueidan C, Ruibal C, de Hoog GS, Gorbushina AA, Untereiner WA, Lutzoni F (2008) A rock-inhabiting ancestor for mutualistic and pathogen-rich fungal lineages. Stud Mycol 61:111–119
Gueidan C, Roux C, Lutzoni F (2007) Using a multigene analysis to assess generic delineation and character evolution in Verrucariaceae (Verrucariales, Ascomycota). Mycol Res 111:1147–1170
Haase G, Sonntag L, Melzer-Krick B, De Hoog GS (1999) Phylogenetic inference by SSU-gene analysis of members of the Herpotrichiellaceae with special reference to human pathogenic species. Stud Mycol 43:80–97
Hambleton S, Tsuneda A, Currah RS (2003) Comparative morphology and phylogenetic placement of two microsclerotial black fungi from Sphagnum. Mycologia 95:959–975
Harris RC (1995) More Florida Lichens. Privately published, New York
Hibbett DS, Binder M, Bischoff JF, Blackwell M, Cannon PF, Eriksson OE, Huhndorf S, James T, Kirk PM, Lücking R, Lumbsch HT, Lutzoni F, Matheny PB, McLaughlin DJ, Powell MJ, Redhead S, Schoch CL, Spatafora JW, Stalpers JA, Vilgalys R, Aime MC, Aptroot A, Bauer R, Begerow D, Benny GL, Castlebury LA, Crous PW, Dai YC, Gams W, Geiser DM, Griffith GW, Gueidan C, Hawksworth DL, Hestmark G, Hosaka K, Humber RA, Hyde KD, Ironside JE, Kõljalg U, Kurtzman CP, Larsson KH, Lichtwardt R, Longcore J, Miadlikowska J, Miller A, Moncalvo JM, Mozley-Standridge S, Oberwinkler F, Parmasto E, Reeb V, Rogers JD, Roux C, Ryvarden L, Sampaio JP, Schüßler A, Sugiyama J, Thorn RG, Tibell L, Untereiner WA, Walker C, Wang Z, Weir A, Weiss M, White MM, Winka K, Yao YJ, Zhang N (2007) A higher-level phylogenetic classification of the fungi. Mycol Res 111:509–547
Hyde KD, Gareth Jones EB, Liu J-K, Ariyawansa H, Boehm E, Boonmee S, Braun U, Chomnunti P, Crous PW, Dai D-Q, Diederich P, Dissanayake A, Doilom M, Doveri F, Hongsanan S, Jayawardena R, Lawrey JD, Li Y-M, Liu Y-X, Lücking R, Monkai J, Muggia L, Nelsen MP, Pang K-L, Phookamsak R, Senanayake IC, Shearer CA, Suetrong S, Tanaka K, Thambugala KM, Wijayawardene NN, Wikee S, Wu H-X, Zhang Y, Aguirre-Hudson B, Alias SA, Aptroot A, Bahkali AH, Bezerra JL, Bhat DJ, Camporesi E, Chukeatirote E, Gueidan C, Hawksworth DL, Hirayama K, De Hoog GS, Kang J-C, Knudsen K, Li W-J, Li X-H, Liu Z-Y, Mapook A, McKenzie EHC, Miller AN, Mortimer PE, Phillips AJL, Raja HA, Scheuer C, Schumm F, Taylor JE, Tian Q, Tibpromma S, Wanasinghe DN, Wang Y, Xu J-C, Yacharoen S, Yan J-Y, Zhang M (2013) Families of Dothideomycetes. Fungal Divers 63:1–313
Inderbitzin P, Lim SR, Volkmann-Kohlmeyer B, Kohlmeyer J, Berbee ML (2004) The phylogenetic position of Spathulospora based on DNA sequences from dried herbarium material. Mycol Res 108:737–748
James TY, Kauff F, Schoch C, Matheny PB, Hofstetter V, Cox CJ, Celio G, Gueidan C, Fraker E, Miadlikowska J, Lumbsch T, Rauhut A, Reeb V, Arnold AE, Amtoft A, Stajich JE, Hosaka K, Sung GH, Johnson D, O’Rourke B, Binder M, Curtis JM, Slot JC, Wang Z, Wilson AW, Schüßler A, Longcore JE, O’Donnell K, Mozley-Standridge K, Porter D, Letcher PM, Powell MJ, Taylor JW, White MM, Griffith GW, Davies DR, Sugiyama J, Rossman AY, Rogers JD, Pfister DH, Hewitt D, Hansen K, Hambleton S, Shoemaker RA, Kohlmeyer J, Volkmann-Kohlmeyer B, Spotts RA, Serdani M, Crous PW, Hughes KW, Matsuura K, Langer E, Langer G, Untereiner WA, Lücking R, Büdel B, Geiser DM, Aptroot A, Buck WR, Cole MS, Diederich P, Printzen C, Schmitt I, Schultz M, Yahr R, Zavarzin A, Hibbett DS, Lutzoni F, McLaughlin DJ, Spatafora JW, Vilgalys R (2006) Reconstructing the early evolution of the fungi using a six-gene phylogeny. Nature 443:818–822
Karuppayil SM, Szaniszlo PJ (1997) Importance of calcium to the regulation of polymorphism in Wangiella dermatitidis. J Med Vet Mycol 35:379–388
Kost G (1988) Moss inhabiting basidiomycetes: interactions between basidiomycetes and Bryophyta. Endocytobiosis Cell Res 5:287–308
Le Calvez T, Burgaud G, Mahé S, Barbier G, Vandenkoornhuyse P (2010) Fungal Divers in Deep-Sea Hydrothermal Ecosystems. Appl Environ Microbiol 75:6415–6421
Little AEF, Currie CR (2007) Symbiotic complexity: discovery of a fifth symbiont in the attine ant–microbe symbiosis. Biol Lett 3:501–504
Liu YJ, Hall BD (2004) Body plan evolution of ascomycetes, as inferred from an RNA polymerase II phylogeny. Proc Natl Acad Sci U S A 101:4507–4512
Liu H, Kauffman S, Becker JM, Szaniszlo PJ (2004) Wangiella (Exophiala) dermatitidis, WdChs5p, a class V chitin synthase is essential for sustained cell growth at temperature of infection. Eukaryot Cell 3:40–51
Lumbsch HT, Schmitt I, Lindemuth R, Miller A, Mangold A, Fernandez F, Huhndorf S (2005) Performance of four ribosomal DNA regions to infer higher-level phylogenetic relationships of inoperculate euascomycetes (Leotiomycota). Mol Phylogenet Evol 34:512–524
Luttrell ES (1951) Taxonomy of pyrenomycetes. Univ Mo Stud Sci Ser 24:1–120
Luttrell ES (1955) The ascostromatic Ascomycetes. Mycologia 47:511–532
Lutzoni F, Wagner P, Reeb V, Zoller S (2000) Integrating ambiguously aligned regions of DNA sequences in phylogenetic analyses without violating positional homology. Syst Biol 49:628–651
Lutzoni F, Pagel M, Reeb V (2001) Major fungal lineages are derived from lichen symbiotic ancestors. Nature 411:937–940
Lutzoni F, Kauff F, Cox C, McLaughlin D, Celio G, Dentinger B, Padamsee M, Hibbett D, James TY, Baloch E, Grube M, Reeb V, Hofstetter V, Schoch C, Arnold AE, Miadlikowska J, Spatafora J, Johnson D, Hambleton S, Crockett M, Shoemaker R, Sung GH, Lücking R, Lumbsch T, O'Donnell K, Binder M, Diederich P, Ertz D, Gueidan C, Hansen K, Harris RC, Hosaka K, Lim YW, Matheny B, Nishida H, Pfister D, Rogers J, Rossman A, Schmitt I, Sipman H, Stone J, Sugiyama J, Yahr R, Vilgalys R (2004) Assembling the fungal tree of life: progress, classification, and evolution of subcellular traits. Am J Bot 91:1446–1480
Maddison WP, Maddison DR (2003) MacClade: analysis of phylogeny and character evolution, Version 4.6. Sinauer. Sunderland, Massachusetts
Mason-Gamer R, Kellogg E (1996) Testing for phylogenetic conflict among molecular datasets in the tribe Triticeae (Graminae). Syst Biol 45:524–545
Mayer VE, Volgmayr H (2009) Mycelial carton galleries of Azteca brevis (Formicidae) as a multi-species network. Proc R Soc B Biol Sci 276:3265–3273
Mendoza L, Karuppayil SM, Szaniszlo PJ (1993) Calcium regulates in vitro dimorphism in chromoblastomycotic fungi. Mycoses 36:157–164
Miller MA, Pfeiffer W, Schwartz T (2010) Creating the CIPRES Science Gateway for inference of large phylogenetic trees. Proceedings of the Gateway Computing Environments Workshop (GCE), New Orleans, 1–8
Posada D, Crandall KA (1998) Modeltest: testing the model of DNA substitution. Bioinformatics 14:817–818
Prenafeta-Boldú FX, Summerbell R, de Hoog GS (2006) Hydrocarbons as their sole carbon and energy source. Mycol Res 105:477–484
Prenafeta-Boldú FX, Kuhn A, Luykx D, Anke H, van Groenestijn JW, de Bont JAM (2001) Fungi growing on aromatic hydrocarbons: biotechnology’s unexpected encounter with biohazard? FEMS Microbiol Rev 30:109–130
Racovitza A (1959) Étude systématique et biologique des champignons bryophiles. Mém Mus Nat Hist Nat Sér B Bot 10:1–288
Réblová M, Untereiner WA, Réblová K (2013) Novel evolutionary lineages revealed in the Chaetothyriales (Fungi) based on multigene phylogenetic analyses and comparison of ITS Secondary Structure. PLoS One 8:e63547
Ronquist F, Teslenko M, van der Mark P, Ayres D, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2011) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542
Rossman AY, Schoch CL, Farr DF, Nishijima K, Keith L, Goenaga R (2010) Dolabra nepheliae on rambutan and lychee represents a novel lineage of phytopathogenic Eurotiomycetes. Mycoscience 51:300–309
Ruibal C, Platas G, Bills GF (2005) Isolation and characterization of melanised fungi from limestone formations in Mallorca. Mycol Prog 4:23–38
Ruibal C, Platas G, Bills GF (2008) High diversity and morphological convergence among melanised fungi from rock formations in the Central Mountain System of Spain. Persoonia 21:93–110
Schnitzler N, Peltroche-Llacsahuanga H, Bestier N, Zijndorf J, Lütticken R, Haase G (1999) Effect of melanin and carotenoids of Exophiala (Wangiella) dermatitidis on phagocytosis, oxidative burst, and killing by human neutrophils. Infect Immun 67:94–101
Schoch C, Sung GH, López-Giráldez F, Townsend JP, Miadlikowska J, Hofstetter V, Robbertse B, Matheny B, Kauff F, Wang Z, Gueidan C, Andrie RM, Trippe K, Ciufetti LM, Wynns A, Fraker E, Hodkinson BP, Bonito G, Groenewald JZ, Arzanlou M, De Hoog GS, Crous PW, Hewitt D, Pfister D, Peterson K, Gryzenhout M, Wingfield MJ, Aptroot A, Suh SO, Blackwell M, Hillis DM, Griffith GW, Castlebury LA, Rossman A, Lumbsch HT, Lücking R, Büdel B, Rauhut A, Diederich P, Ertz D, Geiser DM, Hosaka K, Inderbitzin P, Kohlmeyer J, Volkmann-Kohlmeyer B, Mostert L, O'Donnell K, Sipman H, Rogers JD, Shoemaker R, Sugiyama J, Summerbell RC, Untereiner WA, Johnston PR, Stenroos S, Zuccaro A, Dyer PS, Crittenden PD, Cole MS, Hansen K, Trappe JM, Yahr R, Lutzoni F, Sapatafora JW (2009) The Ascomycota tree of life: a phylum-wide phylogeny clarifies the origin and evolution of fundamental reproductive and ecological traits. Syst Biol 58:224–239
Seyedmousavi S, Badali H, Chlebicki A, Zhao J, Prenafeta-Boldú FX, De Hoog GS (2011) Exophiala sideris, a novel black yeast isolated from environments polluted with toxic alkyl benzenes and arsenic. Fungal Biol 115:1030–1037
Spatafora JW, Schoch CL, Johnson D, Sung G, Hosaka K, O’Rourke B, Serdani M, Spotts R (2006) A five-gene phylogenetic analysis of the Pezizomycotina. Mycologia 98:1018–1028
Spatafora JW, Mitchell TG, Vilgalys R (1995) Analysis of genes coding for small-subunit rRNA sequences in studying phylogenetics of Dematiaceous fungal pathogens. J Clin Microbiol 33:1322–1326
Stamatakis A, Hoover P, Rougemont J (2008) A rapid bootstrap algorithm for the RAxML web-servers. Syst Biol 75:758–771
Stamatakis A, Ludwig T, Meier H (2005) A fast program for maximum likelihood-based inference of large phylogenetic trees. Bioinformatics 21:456–463
Stenroos S, Laukka T, Huhtinen S, Döbbeler P, Myllys L, Syrjänen K, Hyvönen J (2010) Multiple origins of symbioses between ascomycetes and bryophytes suggested by a five-gene phylogeny. Cladistics 26:281–300
Sterflinger K, de Hoog GS, Haase G (1999) Phylogeny and ecology of meristematic ascomycetes. Stud Mycol 43:5–22
Swofford DL (1999) PAUP*: phylogenetic analysis using parsimony (* and other methods) Version 4.0b10. Sinauer, Sunderland
Tibell L (1984) A reappraisal of the taxonomy of Caliciales. In: Hertel H, Oberwinkler F (eds), Beiträge zur Lichenologie. Festschrift J. Poelt. Beiheft zur Nova Hedwigia 79. J. Cramer, Vaduz, pp 597–713
Tibell L, Wedin M (2000) Mycocaliciales, a new order for nonlichenised calicioid fungi. Mycologia 92:577–581
Tsuneda A, Hambleton S, Currah RS (2011) The anamorph genus Knufia and its phylogenetically allied species in Coniosporium, Sarcinomyces, and Phaeococcomyces. Botany 89:523–536
Untereiner WA (1997) Taxonomy of selected members of the ascomycete genus Capronia with notes on anamorph-teleomorph connections. Mycologia 89:120–131
Untereiner WA (2000) Capronia and its anamorphs: exploring the value of morphological and molecular characters in the systematics of the Herpotrichiellaceae. Stud Mycol 45:141–149
Untereiner WA, Gueidan C, Orr M-J, Diederich P (2011) The phylogenetic position of the lichenicolous ascomycete Capronia peltigerae. Fungal Divers 49:225–233
Voglmayr H, Mayer V, Maschwitz U, Moog J, Djieto-Lordon C, Blatrix R (2011) The diversity of ant-associated black yeasts: insights into a newly discovered world of symbiotic interactions. Fungal Biol 115:1077–1091
Wedin M, Tibell L (1997) Phylogeny and evolution of Caliciaceae, Mycocaliciaceae, and Sphinctrinaceae (Ascomycota), with notes on the evolution of the prototunicate ascus. Can J Bot 75:1236–1242
White TJ, Bruns T, Lee S, Taylor JW (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR Protocols: a Guide to Methods and Applications. Academic Press, New York, pp 315–322
Winka K (2000) Phylogenetic relationships within the Ascomycota based on 18S rDNA sequences. PhD thesis, Umeå University, Sweden
Winka K, Eriksson OE, Bång Å (1998) Molecular evidence for recognizing the Chaetothyriales. Mycologia 90:822–830
Acknowledgements
The authors would like to thank Peter Döbbeler for providing a description of the new family Epibryaceae, as well as Putarak Chomnunti and Kevin Hyde for sharing their expertise on Chaetothyriaceae and Trichomeriaceae. This research was supported by internal funding for molecular work from the Natural History Museum in London.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gueidan, C., Aptroot, A., da Silva Cáceres, M.E. et al. A reappraisal of orders and families within the subclass Chaetothyriomycetidae (Eurotiomycetes, Ascomycota). Mycol Progress 13, 990 (2014). https://doi.org/10.1007/s11557-014-0990-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11557-014-0990-2