Abstract
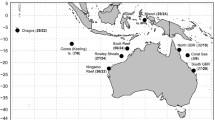
Genetic variability and structuring of rabbitfish populations with contrasting life histories, Siganus argenteus and Siganus fuscescens were determined using allozyme analysis. A total of 13–14 polymorphic loci were examined from samples collected in 2002 and 2003 from eight reefs representing 25 populations north (Kuroshio Current) and south (Mindanao Current) of the bifurcation of the North Equatorial Current along the eastern Philippine coast. S. fuscescens populations (H OBS = 0.085) showed higher heterozygosity than S. argenteus (H OBS = 0.053), consistent with predictions of the neutral theory for demersal egg spawners compared to pelagic egg spawners. The generally lower genetic variability of Kuroshio populations may be due to greater environmental disturbance affecting larval mortality and recruitment success. There was no significant overall population genetic structuring for S. argenteus (F ST = 0.01485, P > 0.05) compared to S. fuscescens (F ST = 0.03275, P < 0.05). The latter species showed highly significant genetic structuring among Kuroshio and Mindanao Current populations in both 2002 and 2003 (F CT = 0.08120, P < 0.05; F CT = 0.07500, P < 0.05, respectively), as well as among populations within regions. This conforms to expectations of correlations between observed population genetic structure and life history features related to dispersal potential and gene flow. However, there were significant temporal (i.e., 2002 vs. 2003 samples) genetic variations for both S. fuscescens (F CT = 0.08542, P < 0.05) and S. argenteus (F CT = 0.06330, P < 0.05), which may reflect interannual variability in recruitment success. Differences in population spatial genetic patterns between the two reef fish species suggest that broad scale physical factors (e.g. NEC bifurcation) and regional environmental perturbations (e.g. incidence of typhoons) affect population genetic structure of sympatric congeneric species with different life histories differently. Finer scale ecological processes, which affect larval dispersal and recruitment (e.g., local hydrographic features, distribution of habitats), particularly in the Mindanao Current region, exert more influence on structuring populations of S. fuscescens.



Similar content being viewed by others
References
Aliño PM, Gomez ED (1994) Philippine coral reef conservation: its significance to the South China sea. In: Yamazato K, Ishijima S, Sakihara S, Taira H, Shimabukuro Z, Teruya F, Nishihara F (eds) Development and conservation in the Asia Pacific Region. Proceedings of the regional conference for East–West Center Association Okinawa, Japan pp 222–229
Avise N (1994) Molecular markers, natural history and evolution. Chapman-Hall, New York
Bay LK, Crozier RH, Caley MJ (2006) The relationship between population genetic structure and pelagic larval duration in coral reef fishes on the Great Barrier Reef. Mar Biol 149:1247–1256
Belkhir K, Bonhomme F, Borsa P, Mathieu E, Roux M (1993) Genetix: Logiciel d’analyse des donees du groupe de genetique des populations de Montpellier. Ver 01, Universite Montpellier II, France
Bell JL, Moyer TJ, Numachi K (1982) Morphological and genetic variation in Japanese populations of the anemonefish Amphiprion clarkii. Mar Biol 72:99–108
Bernardi G (2000) Barriers to gene flow in Embiotoca jacksoni, a marine fish lacking a pelagic larval stage. Evolution 54:226–237
Bernardi G, Holbrook SJ, Schmitt RJ (2001) Gene flow at three spatial scales in a coral reef fish, the threespot dascyllus, Dascyllus trimaculatus. Mar Biol 138:457–465
Bonhomme F, Planes S (2000) Some evolutionary arguments about what maintains the pelagic interval in reef fishes. Environ Biol Fishes 59:365–383
Buonaccorsi VP, Westerman M, Stannard J, Kimbrell C, Lynn E, Vetter RD (2004) Molecular genetic structure suggests limited larval dispersal in grass rockfish, Sebastes rastrelliger. Mar Biol 145:779–788
Buonaccorsi VP, Kimbrell C, Lynn E, Vetter RD (2005) Limited larval dispersal and introgressive hybridization influence genetic structure and conservation strategies for brown rockfish, Sebastes auriculatus. Conserv Genet 6:697–713
Burton RS, Lee BN, (1994) Nuclear and mitochondrial gene geneologies and allozyme polymorphism across a major phylogeographic break in the copepod Tigriopus californicus. Proc Natl Acad Sci U S A 91:5197–5201
Caley MJ, Carr MH, Hixon MA, Hughes TP, Jones GP, Menge BA (1996) Recruitment and the local dynamics of open marine populations. Annu Rev Ecol Syst 27:477–500
Davis BJ, de Martini EE, McGee K (1981) Gene flow among populations of a teleost (painted greenling, Oxylebius pictus) from Puget Sound to southern California. Mar Biol 65:17–23
Dawson MN (2001) Phylogeography in coastal marine animals: a solution from California? J Biogeogr 28:723–736
Dawson MN, Louie KD, Barlow M, Jacobs DK, Swift CC (2002) Comparative phylogeography of sympatric sister species. Clevelandia ios and Eucyclogobius newberryi (Teleostei, Gobiidae), across the California transition zone. Mol Ecol 11:1065–1075
Dela Paz R, Aragones N (1990) Ecological notes on the juvenile siganid (padas) harvest in Bolinao, Pangasinan, Philippines. Micronesia 22(2):25–29
Doherty P, Mather P, Planes S (1994) Acanthochromis polyacanthus, a fish lacking larval dispersal, has genetically differentiated populations at local and regional scales on the Great Barrier Reef. Mar Biol 121:11–21
Doherty PJ, Planes S, Mather P (1995) Gene flow and larval duration in seven species of fish from the Great Barrier Reefs. Ecology 76:2373–2391
Exadactylos A, Geffren AJ, Panagiotaki P, Thorpe JP (2003) Population structure of dover sole Solea solea: RAPD and allozyme data indicate divergence in European stocks. Mar Ecol Prog Ser 246:253–264
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genetics 131:479–491
Fauvelot C, Planes S (2002) Understanding origins of present-day genetic structure in marine fish: biologically or historically driven patterns? Mar Biol 141:773–788
Harrison PL, Wallace CC (1990) Reproduction, dispersal, and recruitment of scleractinian coral. In: Dubinsky Z (ed) Ecosystems of the world, vol 25: coral Reefs. Elsevier, pp 133–208
Hellberg ME (2007) Footprints on water: the genetic wake of dispersal among reefs. Coral Reefs doi:10.1007/s00338-007-0205-2
Johnson MS, Watts RJ, Black R (1994) High levels of genetic subdivision in peripherally isolated populations of the atherinid fish Craterocephalus capreoli in the Houtman Abrolhos Islands, Western Australia. Mar Biol 119:179–184
Jones GP, Milicich MJ, Emslie MJ, Lunow C (1999) Self-recruitment in a coral reef fish population. Nature 402:802–804
Juinio-Menez MA, Magsino RM, Ravago RG, Yu ET (2003) Genetic structure of Linckia laevigata and Tridacna crocea among the palawan shelf and shoal reefs. Mar Biol 142:717–726
Lacson JM (1992) Minimal genetic variation among samples of six species of coral reef fishes at La Parguera, Puerto Rico and Discovery Bay, Jamaica. Mar Biol 112:327–331
Leis JM (1991) The pelagic stage of reef fishes: the larval biology of coral reef fishes. In: Sale PF (ed) The ecology of fishes on coral reefs, Academic Press, San Diego, pp 183–230
Magsino RM, Juinio-Menez MA, Planes S (2002) Preliminary analysis on the genetic variation of Tridacna crocea among seven reefs in the eastern Philippines. UPV J Nat Sci 1/2:81–89
Magsino RM, Juinio-Menez MA (unpubl. data) Morphological variation in three populations of Siganus argenteus and Siganus fuscescens along the eastern Philippine coasts. Paper presented at the 10th Southern Luzon Zonal Center Research and Development Review, Ateneo de Manila University, Manila, Philippines, 10–11 November 2006
Mantel N (1967) The detection of disease clustering and a generalized regression approach. Cancer Res 27:209–220
Miller M (1997) Tools for population genetics analyses (TFPGA) 1.3. A Windows program for the analysis of allozyme and molecular population genetic data. Software distributed by the author
Muss A, Robertson DR, Stepien CA, Wirtz P, Bowen B (2001) Phylogeography of Ophioblennius: the role of oceanic currents and geography in reef fish evolution. Evolution 55(3):561–572
Ochavillo DG (2001) Evidences for self-recruitment in a tropical coral reef fish population. PhD dissertation, University of Southern California, USA
Planes S, Doherty PJ (1997a) Genetic relationships of the colour morphs of Acanthochromis polyacanthus (Pomacentridae) on the northern Great Barrier Reef. Mar Biol 130:109–117
Planes S, Doherty PJ (1997b) Genetic and color interaction at a contact zone of Acanthochromis polyacanthus: a marine fish lacking pelagic larvae. Evolution
Planes S, Doherty PJ, Bernardi G (2001) Strong genetic divergence among populations of a marine fish with limited dispersal, Acanthochromis polyacanthus, within the Great Barrier Reef and the Coral Sea. Evolution 55(11):2263–2273
Purcell JFH, Cowen RK, Hughes CR, Williams DA (2006) Weak genetic structure indicates strong dispersal limits: a tale of two coral reef fish. Proc R Soc 273:1483–1490
Qui B, Lukas R (1996) Seasonal and interannual variability of the North Equatorial Current, the Mindanao Current and the Kuroshio along the pacific western boundary. J Geophys Res 101:12315–12330
Ravago-Gotanco RG, Magsino RM, Juinio-Menez MA (2007) Influence of the North Equatorial Current on the population genetic structure of Tridacna crocea (Mollusca:Tridacnidae) along the eastern Philippine coast. Mar Ecol Prog Ser 336:161–168
Raymond M, Rousset F (1995) GENEPOP (version 1.2): a population genetics software for exact tests and ecumenism. J Hered 86:248–249
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225
Riginos C, Nachman MW (2001) Population subdivision in marine environments: the contributions of biogeography, geographical distance, and discontinuous habitat to genetic differentiation in a blennioid fish, Axoclinus nigricaudus. Mol Ecol 10:1439–1453
Riginos C, Victor BC (2001) Larval spatial distributions and other early life-history characteristics predict genetic differentiation in eastern Pacific blennioid fishes. Proc R Soc Bull 268:1931–1936
Rollon RN, Samson MS, Vergara MWB (2003) Estimating biomass from the cover of Gelidiella acerosa along the coasts of Eastern Philippines. Botanica Marina 46:497–502
Schneider S, Roessli D, Excoffier L (2000) ARLEQUIN ver. 2.000: a software for population genetics data analysis. Genetics and biometry laboratory. University of Geneva, Switzerland
Selkoe KA, Gaines SD, Caselle JE, Warner RR (2006) Current shifts and kin aggregation explain genetic patchiness in fish recruits. Ecology 87(12):3082–3094
Shaw CR, Prasad R (1970) Starch gel electrophoresis of enzymes—a compilation of recipes. Biochem Genet 4:297–320
Shokita S, Kakazu K, Tomori A, Toma T (1991) Aquaculture in tropical areas, Rabbitfishes (Siganus spp). Midoro Shobo Co. Ltd., pp 360
Shulman MJ, Bermingham E (1995) Early life histories, ocean currents, and the population genetics of Caribbean reef fishes. Evolution 49:897–910
Sugimoto T, Kimura S, Tadokoro K (2001) Impact of El Niño events and climate regime shift on living resources in the western North Pacific. Progr Oceanogr 49:113–127
Swearer SE, Caselle JE, Lea D, Warner RR (1999) Larval retention and recruitment in an island population of a coral reef fish. Nature 402:799–802
Taylor MS, Hellberg ME (2003) Genetic evidence for local retention of pelagic larvae in a Caribbean reef fish. Science 299:107–109
Terry A, Bucciarelli G, Bernardi G (2000) Restricted gene flow and incipient speciation in disjunct Pacific Ocean and Sea of Cortez populations of a reef fish species, Girella nigricans. Evolution 54:652–659
Waples RS, Rosenblatt RH (1987) Patterns of larval drift in Southern California marine shore fishes inferred from allozyme data. Fish Bull US 85:1–11
Ward RD (1989) Molecular population genetics of marine organisms. In: Reproduction, genetics and distribution of marine organisms. In: Ryland JS, Tyler PA, (ed) 23rd European Marine Biology Symposium, Olsen and Olsen, Fredensburg, pp 235–249
Weir BS (1990) Genetic data analysis. Sinauer, Sunderland
Weir BS, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370
Woodland DJ (1990) Revision of the fish family Siganidae with descriptions of two new species and comments on distribution and biology. Indo-Pacific fishes 19:136pp Bishop Museum, Honolulu, Hawaii, USA
Acknowledgments
This study is a component of the Pacific seaboard Research and Development Program funded by the Department of Science and Technology–Philippine Council for Marine and Aquatic Research and Development. RMM would like to thank the Philippine Commission for Higher Education (CHED) for the thesis grant and various people who helped in the collection of fish samples. The authors are grateful for the invaluable assistance of R. Ravago-Gotanco (UPMSI) in improving the analysis and discussion of the results. The experiments performed in this study comply with the current laws of the Republic of the Philippines. This is contribution number 365 of the UP Marine Science Institute.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by A. McLachlan.
Rights and permissions
About this article
Cite this article
Magsino, R.M., Juinio-Meñez, M.A. The influence of contrasting life history traits and oceanic processes on genetic structuring of rabbitfish populations Siganus argenteus and Siganus fuscescens along the eastern Philippine coasts. Mar Biol 154, 519–532 (2008). https://doi.org/10.1007/s00227-008-0946-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00227-008-0946-7